Headline numbers
The most useful comparison is between the largest published trial for each medication, run in adults living with obesity who didn’t have type 2 diabetes.
| Feature |
Retatrutide |
Wegovy (semaglutide) |
| How it works |
Triple receptor agonist (GLP-1, GIP, glucagon) |
GLP-1 receptor agonist (single hormone) |
| Highest-dose weight loss in obesity (no type 2 diabetes) |
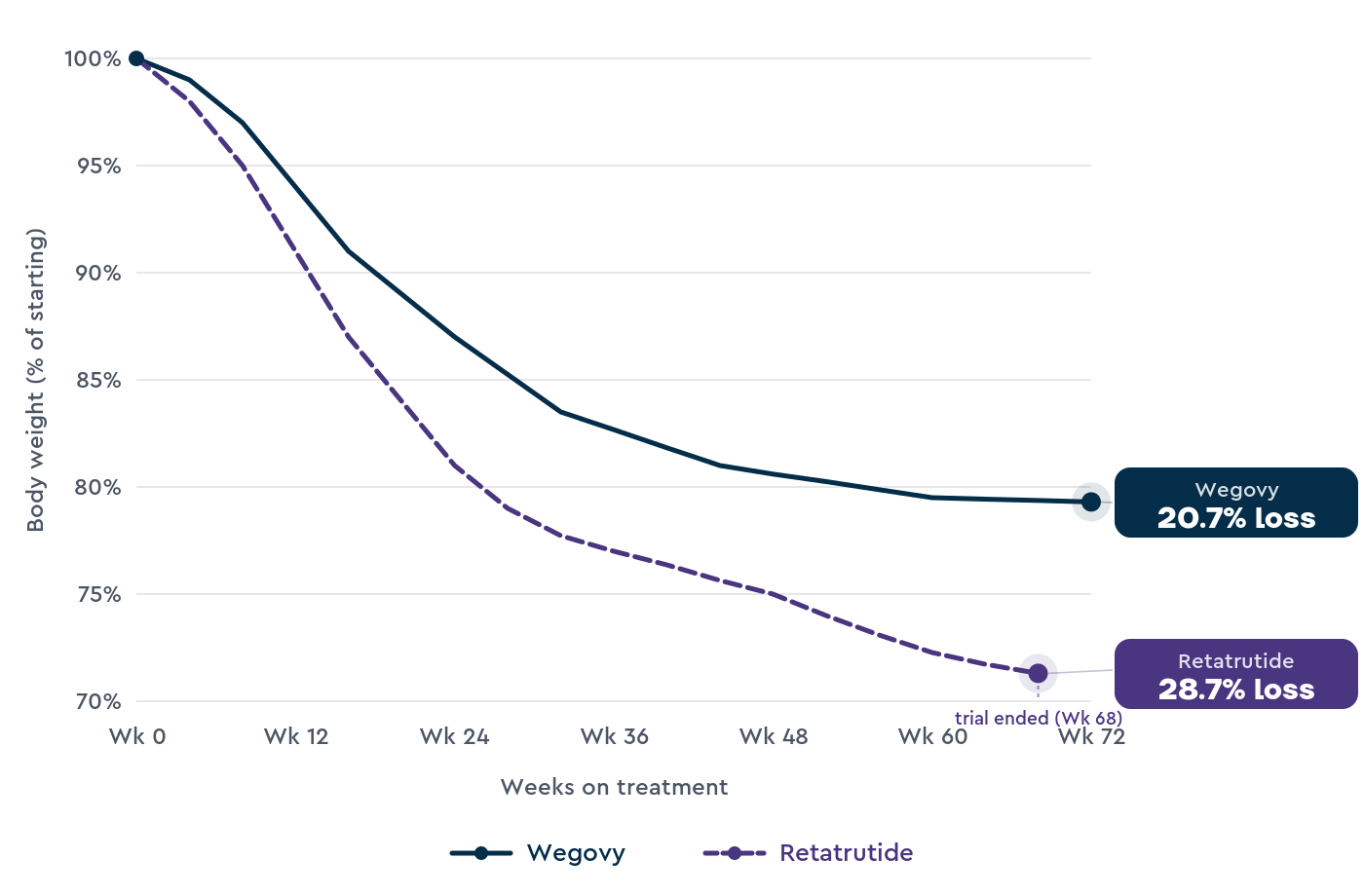
28.7% at 68 weeks, 12 mg dose (TRIUMPH-4, 445 participants)1 |
20.7% at 72 weeks, 7.2 mg dose (STEP-UP, 1,742 participants)3; 14.9% at 68 weeks, 2.4 mg dose (STEP 1, 1,961 participants)2 |
| Administration |
Once a week, subcutaneous injection (in trials) |
Once a week, subcutaneous injection |
| UK approval |
Not approved; Phase 3 human trials ongoing |
MHRA Sep 2021 (2.4 mg) and Jan 2026 (7.2 mg); NICE TA875 Mar 2023 |
| Cardiovascular outcomes data |
No completed cardiovascular outcomes trial |
20% reduction in serious heart events (SELECT trial, 17,604 participants)4 |
| UK availability in 2026 |
Not legally available outside clinical trials |
UK pharmacy with prescription; NHS specialist services; private programmes |
| Real-world safety record |
Trial data only |
Years of real-world safety data |
The 28.7% figure for retatrutide comes from TRIUMPH-4, a Phase 3 trial of 445 adults living with obesity and knee osteoarthritis, reported by Lilly in a December 2025 press release.1
The full TRIUMPH-4 paper hasn’t yet been published in a peer-reviewed journal.
Wegovy’s higher 7.2 mg dose was MHRA-approved in January 2026. The 20.7% figure comes from the STEP-UP trial of 1,742 adults living with obesity, published in NEJM in 2025.3
The 14.9% figure for Wegovy 2.4 mg comes from STEP 1, the registration trial of 1,961 adults living with obesity but no type 2 diabetes, published in NEJM in 2021.2
These are different trials, different populations, and different time points, so the comparison is approximate.
There’s no trial directly comparing retatrutide and Wegovy, and there isn’t likely to be one before retatrutide is approved.
How retatrutide and Wegovy work
Both medications work on the body’s appetite-regulating hormones, but they target different ones, which is why the trial results differ.
How Wegovy works
Wegovy contains semaglutide, a GLP-1 receptor agonist.
It mimics GLP-1, a gut hormone the body releases after eating that reduces appetite, slows stomach emptying, and signals the brain that we’re full.2
In practice, people on Wegovy often report reduced food noise, comfortably eating smaller portions, and less interest in snacking between meals.
The standard maintenance dose is 2.4 mg once a week, with the higher 7.2 mg dose now also approved as of January 2026.
How retatrutide works
Retatrutide is a triple receptor agonist.
It mimics three hormones: GLP-1 (the same as Wegovy), GIP (a related gut hormone that affects insulin and fat storage, the same as Mounjaro), and glucagon.5
The GLP-1 and GIP components reduce appetite and improve insulin sensitivity.
The third hormone, glucagon, is what makes retatrutide different. Glucagon raises the body’s resting energy use and helps it use stored fat for fuel.5
That third pathway is why retatrutide is called a triple agonist rather than a dual agonist like Mounjaro, and why its trial weight-loss numbers are higher than those of single- or dual-action medications.
Why retatrutide produces more weight loss
Wegovy works mainly by reducing how much food you eat. Retatrutide does that too, and it also raises the energy our body uses at rest.
Adding the GIP component improves insulin sensitivity and may help the body tolerate higher doses of the GLP-1 component. Adding the glucagon component raises resting energy use.
In the Phase 2 trial, weight loss continued at 48 weeks across all dose groups, suggesting retatrutide may not plateau as single-receptor medications often do.5
This explains why TRIUMPH-4 produced 28.7% weight loss at 68 weeks while STEP-UP produced 20.7% at 72 weeks on Wegovy 7.2 mg, and STEP 1 produced 14.9% at 68 weeks on Wegovy 2.4 mg.1,2,3
The caveat is that we’re comparing trials that weren’t designed to be compared.
TRIUMPH-4 enrolled adults living with obesity and knee osteoarthritis, with a starting body mass index (BMI) averaging 40.4.
STEP 1 and STEP-UP enrolled adults living with obesity, with slightly lower starting BMIs and different inclusion criteria.
Trial protocols, food advice, and exercise guidance also differed across the studies.
Still, given the triple-action of retatrutide, we would expect it to lead to more weight loss. But as the two drugs haven’t been compared directly under the same conditions, we can’t know for sure.
Side effects compared
The two medications share most of their side-effect profile because the GLP-1 component drives most of the nausea, stomach, and gut symptoms, and how these settle as the body adjusts.
Common side effects on Wegovy
The most frequent side effects in STEP 1 were stomach and gut symptoms: nausea, diarrhoea, vomiting, and constipation.2
These were typically mild to moderate and most common when the dose increased. Headache, fatigue, and indigestion were also reported.
Most people who experienced side effects saw them resolve as the dose stabilised over the first few months.
Wegovy has been on the UK market since 2022, which means there are now several years of real-world safety data on top of the trial results.
Common side effects of retatrutide
Retatrutide has a similar side effect profile as Wegovy and other GLP-1 medications, like Mounjaro.
In the Phase 2 trial, nausea ranged from approximately 14% at the 1 mg dose to 60% at the 12 mg dose, with most cases occurring during dose stepping.5 Diarrhoea, vomiting, and constipation followed similar patterns.
In TRIUMPH-4, 18.2% of people on the 12 mg dose stopped because of side effects, compared with around 7% on Wegovy in STEP 1.1,2
Retatrutide trials have also flagged some concerns that aren’t typical for Wegovy.
Heart rate increased by an average of around 6.7 beats per minute on the 12 mg dose, peaking at about week 24 and easing slightly afterwards.5
Irregular heartbeats were reported in 2 to 11% of people on retatrutide compared with around 2% on placebo, with none classified as serious.5
Dysesthesia (a skin sensation often described as tingling, numbness, or burning) affected around 21% of people on the 12 mg dose in TRIUMPH-4.1 This concern emerged in Phase 3 and wasn’t seen in Phase 2.
Until Phase 3 is complete and real-world data starts to accumulate, retatrutide’s full safety profile won’t be clear.
That’s a real difference compared with Wegovy, where real-world data has been accumulating for years.
Cardiovascular and long-term safety
What SELECT showed for Wegovy
The SELECT trial enrolled 17,604 adults with established cardiovascular disease and a body mass index of 27 or higher, who didn’t have type 2 diabetes.4
Participants were randomly assigned to Wegovy 2.4 mg once a week or placebo, and followed for an average of 39.8 months.
The main result the trial measured was the rate of serious heart events: heart attack, stroke, or death from cardiovascular causes. These occurred in 6.5% of participants on Wegovy and 8.0% on placebo, a 20% relative risk reduction.4
This evidence supported the MHRA’s decision to extend Wegovy’s licence in July 2024 to include reducing the risk of serious heart events in adults living with overweight or obesity and established cardiovascular disease.
Wegovy is the only weight loss medication in the UK that can be prescribed to reduce the risk of cardiovascular disease.
The impact of retatrutide on cardiovascular disease
Retatrutide doesn’t have an equivalent cardiovascular outcomes trial yet.
The TRIUMPH Phase 3 programme is focused on weight, type 2 diabetes, knee osteoarthritis, and obstructive sleep apnoea.
Phase 2 and Phase 3 trials so far haven’t reported an excess of serious heart events on retatrutide, but they were short and weren’t designed to test that question.5
For someone living with established cardiovascular disease, this is a real difference between the two medications.
Wegovy has a positive cardiovascular outcomes trial behind it; retatrutide doesn’t, and won’t for several years.
Muscle and lean mass
Some of the weight lost on any GLP-1 medication is muscle, not fat. Across the published trials, around a quarter of total weight loss has come from lean tissue. At retatrutide’s 28.7% total weight loss, the absolute loss in muscle is larger.
Our muscles play a central role in glucose regulation, accounting for around 70-80% of insulin-stimulated glucose uptake after meals. They also support our day-to-day strength and mobility, which becomes more important as we age.
Three factors are most strongly associated with muscle preservation during weight loss: losing weight slowly, adequate protein intake, and resistance training.
A steady rate of weight loss preserves more lean tissue than rapid weight loss. This might mean increasing your medication dose more gradually and combining medication with sustainable nutrition and habit changes.
Research on adults losing weight suggests a protein intake of 1.2 to 1.6 grams per kg of body weight per day.
For someone weighing 90 kg, this amounts to around 110-145 grams of protein daily, or a serving the size of the palm of your hand in each meal.
Common sources include eggs, Greek yoghurt, chicken, fish, tofu, lentils, beans, and cottage cheese. Including a protein source at each meal helps spread intake across the day.
Resistance training is any exercise that works the muscles against weight or resistance. This includes bodyweight exercises (squats, lunges, press-ups), resistance bands, dumbbells, and weight machines.
Even short sessions a couple of times a week can help to maintain muscle mass during weight loss.
Drug interactions and contraception
Slowed gastric emptying can reduce the body’s ability to absorb medications taken by mouth.
The most clinically relevant examples are the combined oral contraceptive pill, warfarin, and thyroid medication.
Mounjaro’s UK label recommends that women on the combined oral contraceptive pill switch to a non-oral method or use backup contraception for four weeks after starting and four weeks after each dose increase.
Wegovy doesn’t carry the same explicit warning at the standard 2.4 mg dose, but the way the medications work is similar, so any woman of reproductive age starting a GLP-1 medication should speak to her GP about contraception options.
Retatrutide’s effect on oral contraceptive absorption isn’t yet documented in detail.
GLP-1 medications, including Wegovy and retatrutide, aren’t recommended during pregnancy or breastfeeding.
Most clinicians recommend stopping the medication at least two months before trying to conceive.
Mental health
GLP-1 medications have been under regulatory review for possible links to suicidal thoughts and mood changes.
The European Medicines Agency reviewed semaglutide and liraglutide in 2023 and 2024 and concluded that the available evidence didn’t support a causal link.
The FDA reached similar conclusions for tirzepatide and semaglutide.
Retatrutide trials haven’t reported a mood-related concern in Phase 2 or Phase 3.
If you notice low mood, anxiety, or thoughts of self-harm while taking any weight loss medication, contact your GP.
UK access today vs the realistic timeline for retatrutide
How to access Wegovy in the UK
Wegovy is available through three main routes in May 2026:
- Private prescription through a regulated UK clinic, online pharmacy, or programme. Most private routes offer Wegovy as part of a structured weight management service rather than as a standalone medication.
- NHS specialist weight management services. NICE recommends Wegovy for adults with a BMI of at least 35 (or 32.5 in some ethnic groups) and at least one weight-related condition. Access is by referral to a tier 3 or specialist service, and supply has been limited.
- Some integrated care boards have pilot programmes that prescribe Wegovy in primary care, with eligibility criteria that vary by region
Why retatrutide isn’t legally available
Retatrutide doesn’t have a UK marketing authorisation, so no UK pharmacy or licensed clinic can lawfully dispense it.
Phase 3 trials (large human trials with thousands of participants) are still running, and Lilly plans to apply for regulatory approval once the TRIUMPH programme publishes its results across 2026.
Based on the Wegovy and Mounjaro timelines, MHRA approval is plausible in late 2027 to mid 2028, with NICE recommendation following 6 to 18 months later. That puts NHS access at 2029 at the earliest.
Anything sold online in 2026 as ‘retatrutide pens’ isn’t real and should be avoided.
The MHRA has disrupted two UK manufacturing facilities producing counterfeit retatrutide and tirzepatide pens, in October 2025 and February 2026.
We’ve covered this in our guide on buying retatrutide online.
Switching considerations
If you’re already on Wegovy, this comparison may prompt the question of whether to wait for retatrutide.
There’s no medical case for stopping a working medication on the assumption that a stronger one will be available in two or three years.
Stopping a GLP-1 medication often leads to weight regain if you stop suddenly and haven’t developed the habits needed to maintain the weight loss.
In the STEP 4 trial, people who stopped Wegovy regained around two-thirds of the weight they’d lost over the following 48 weeks.2
In SURMOUNT-4, people who stopped Mounjaro regained 14% of their starting weight over a year, retaining only about a third of the weight they had originally lost.6
Retatrutide’s withdrawal data hasn’t been published yet, but it would likely follow a similar pattern if patients stopped the medication suddenly, and didn’t use the time on the medication to develop healthy habits to maintain weight loss in the long term.
If retatrutide is approved in the UK and your healthcare team thinks switching makes sense, that conversation is best had at the point of approval, not now.
By then, the real-world safety data will be richer, NICE will have made an access decision, and pricing and supply will be clearer.
Who should consider which
Retatrutide isn’t really an option in the UK in 2026 for anyone outside a clinical trial.
The realistic comparison is between Wegovy and the other MHRA-approved weight loss medication, Mounjaro (tirzepatide). We’ve written about it in our guide on Wegovy vs Mounjaro.
Wegovy might be a sensible first option if:
- You meet NICE’s eligibility criteria and want a NICE-recommended option with several years of UK real-world data.
- You have established cardiovascular disease and want a medication with a cardiovascular outcomes trial behind it.
- You’re working with a clinician, dietitian, or programme that has experience prescribing and supporting Wegovy.
Retatrutide may become an option in the future if:
- It’s approved by the MHRA and recommended by NICE, which is likely several years away
- You haven’t responded well to existing GLP-1 medications, and a triple agonist offers a different approach worth trying
- The Phase 3 safety data on heart rate, irregular heartbeats, and dysesthesia are reassuring once it’s complete
For most people in the UK, the choice now is between Wegovy and Mounjaro, supported by a structured weight management service.
Frequently asked questions
Is retatrutide better than Wegovy?
In trials, retatrutide produced higher weight loss than Wegovy at the highest dose of each: 28.7% at 68 weeks on retatrutide 12 mg, versus 20.7% at 72 weeks on the newer Wegovy 7.2 mg dose, and 14.9% at 68 weeks on the older Wegovy 2.4 mg dose.1,2,3
‘Better’ depends on your needs, goals, and how you respond to the medications.
Wegovy has cardiovascular outcomes data, MHRA approval, NICE access, and years of real-world safety experience. Retatrutide has none of these yet.
Can I get retatrutide instead of Wegovy in the UK?
No. Retatrutide doesn’t have a UK marketing authorisation in 2026, so no UK pharmacy or licensed clinic can lawfully dispense it.
Wegovy is available privately and through some NHS specialist services. We have a separate guide on whether you can get retatrutide on the NHS.
When will retatrutide be available in the UK?
Lilly plans to apply for regulatory approval once the TRIUMPH Phase 3 programme publishes its results across 2026.
Based on the Wegovy and Mounjaro timelines, MHRA approval is realistic in late 2027 to mid 2028, with NICE recommendation following 6 to 18 months later. NHS access is unlikely before 2029.
Is retatrutide safer than Wegovy?
It’s too early to say. Retatrutide is still in Phase 3 and hasn’t been used in real-world settings after approval.
Trials have flagged some concerns not typical of Wegovy: increased heart rate, irregular heartbeats in 2-11% of participants, and dysesthesia.5
Wegovy has a longer safety record, including positive cardiovascular outcomes data from the SELECT trial.4
Does Wegovy have heart benefits that retatrutide doesn’t?
Yes, currently. The SELECT trial found that Wegovy 2.4 mg reduced serious heart events by 20% in adults living with obesity and established cardiovascular disease who didn’t have type 2 diabetes.4
The MHRA approved Wegovy for this cardiovascular indication in July 2024. Retatrutide has no equivalent outcomes trial completed, and won’t for several years.
Can I switch from Wegovy to retatrutide?
Not legally in the UK in 2026, because retatrutide isn’t approved.
If retatrutide is approved in the future, switching would be a clinical decision based on your individual response, side effects, cardiovascular risk profile, and access.
That conversation is best had with your prescriber once the full Phase 3 safety profile and UK access pathway are clear.
Does retatrutide work for type 2 diabetes the way Wegovy does for weight?
The TRANSCEND-T2D-1 trial reported in March 2026 found 16.8% weight loss and a 2.0% reduction in HbA1c (a marker of average blood sugar) at the 12 mg dose over 40 weeks in adults living with type 2 diabetes.
Wegovy isn’t licensed for type 2 diabetes; the same drug at lower doses is licensed under the brand name Ozempic.
Retatrutide’s eventual licensed indications will depend on which TRIUMPH and TRANSCEND trials produce positive results and which Lilly applies for.
What’s the difference between retatrutide and Mounjaro?
Both are made by Lilly. Mounjaro (tirzepatide) is a dual receptor agonist (GLP-1 and GIP). Retatrutide is a triple receptor agonist (GLP-1, GIP, and glucagon).
The third receptor target is the main reason retatrutide’s trial weight loss numbers are higher. Mounjaro is approved and available in the UK; retatrutide isn’t.
Will retatrutide affect oral contraception?
Slowed gastric emptying on GLP-1 medications can reduce how well oral contraceptives are absorbed. Mounjaro’s UK label recommends switching to a non-oral method or using backup contraception during the first four weeks of each dose change.
Whether retatrutide has the same effect isn’t yet documented in detail. Speak to your GP about contraception options before starting any GLP-1 medication.
Does retatrutide cause muscle loss?
Some of the weight lost on any GLP-1 medication is muscle, not fat.
Three factors protect muscle: losing weight slowly, eating enough protein (a portion the size of the palm of your hand at each meal), and resistance training a couple of times a week.
How much does Wegovy cost in the UK?
Through private clinics and online pharmacies, Wegovy typically costs between £150 and £350 a month, depending on dose and provider.
NHS specialist services dispense Wegovy free of charge for eligible adults. Retatrutide pricing isn’t yet known because it isn’t on the UK market.
Take home message
In Phase 3 trials, retatrutide has produced higher weight loss than Wegovy: 28.7% at 68 weeks on the 12 mg dose, compared with 20.7% at 72 weeks on the newer Wegovy 7.2 mg dose, and 14.9% at 68 weeks on the older 2.4 mg dose.1,2,3
Retatrutide’s triple-action likely explains why it seems to produce more weight loss on average than Wegovy.
Wegovy has been MHRA-approved since September 2021 (2.4 mg) and January 2026 (7.2 mg), NICE-recommended since March 2023, and has a positive cardiovascular outcomes trial behind it.4
Retatrutide has none of these yet, and the realistic UK approval window is late 2027 to mid 2028, with NHS access from 2029 at the earliest.
If you’re choosing now, Wegovy is the realistic option. If retatrutide is approved in the future, that conversation can be had at the point of approval, when the safety profile and access pathway are clearer.
Second Nature’s medication programme combines Wegovy or Mounjaro with structured nutrition guidance from registered dietitians, built around the balanced plate model (half vegetables, quarter protein, quarter complex carbohydrates such as wholegrain bread, brown rice, or oats, plus a serving of fat).
Our peer-reviewed JMIR study of the GLP-1RA-supported programme reported an average weight loss of 19.1% at 12 months among active subscribers, with 77.7% achieving at least 10% weight loss.7
Second Nature's Mounjaro and Wegovy programmes
Second Nature provides Mounjaro or Wegovy as part of our Mounjaro and Wegovy weight-loss programmes.
Why choose Second Nature over other medication providers, assuming you're eligible?
Because peace of mind matters.
We've had the privilege of working with the NHS for over eight years, helping people across the UK take meaningful steps toward a healthier, happier life.
Our programmes are designed to meet people where they are, whether that means support with weight loss through compassionate one-to-one health coaching, or access to the latest weight-loss medications (like Mounjaro and Wegovy) delivered alongside expert care from a multidisciplinary team of doctors, psychologists, dietitians, and personal trainers.
At the heart of everything we do is a simple belief: real, lasting change comes from building better habits, not relying on quick fixes. We're here to support that change every step of the way.
With over a decade of experience, thousands of lives changed, and a long-standing record of delivering programmes used by the NHS, we believe we're the UK's most trusted weight-loss programme.
We hope to offer you something invaluable: peace of mind, and the support you need to take that first step.
References
- Eli Lilly and Company. (2025, December 11). Lilly’s triple agonist, retatrutide, delivered weight loss of up to an average of 71.2 lbs along with substantial relief from osteoarthritis pain in first successful Phase 3 trial (TRIUMPH-4 press release).
- Wilding, J.P.H. et al. (2021). Once-weekly semaglutide in adults with overweight or obesity (STEP 1). New England Journal of Medicine, 384, 989-1002.
- Knop, F.K. et al. (2025). Semaglutide 7.2 mg in adults with obesity (STEP-UP). New England Journal of Medicine.
- Lincoff, A.M. et al. (2023). Semaglutide and cardiovascular outcomes in obesity without diabetes (SELECT). New England Journal of Medicine, 389, 2221-2232.
- Jastreboff, A.M. et al. (2023). Triple-hormone-receptor agonist retatrutide for obesity: a Phase 2 trial. New England Journal of Medicine, 389, 514-526.
- Aronne, L.J. et al. (2024). Continued treatment with tirzepatide for maintenance of weight reduction in adults with obesity (SURMOUNT-4). JAMA, 331(1), 38-48.
- Richards, R., Whitman, M., Wren, G., Campion, P. (2025). A Remotely Delivered GLP-1RA-Supported Specialist Weight Management Program in Adults Living With Obesity: Retrospective Service Evaluation. JMIR Formative Research, 9(1), e72577.