At-a-glance comparison
|
Wegovy pill |
Foundayo |
| Drug |
Oral semaglutide 25 mg |
Orforglipron |
| Drug class |
GLP-1 receptor agonist (peptide) |
GLP-1 receptor agonist (small molecule) |
| Manufacturer |
Novo Nordisk |
Eli Lilly |
| Dose |
25 mg once a day |
Up to 17.2 mg once a day (six dose steps) |
| How to take it |
First thing, empty stomach, max 120 ml water, 30-minute fast after |
Any time of day, with or without food, no water restrictions |
| Average weight loss |
13.6% at 64 weeks (OASIS 4)3 |
11.2% at 72 weeks (ATTAIN-1, 36 mg)4 |
| Common side effects |
Nausea, diarrhoea, vomiting |
Nausea, constipation, diarrhoea, vomiting |
| FDA approval |
22 December 2025 |
1 April 2026 |
| UK status |
MHRA review ongoing; expected late 20261 |
Submission underway; expected 20272 |
What each medication is
The Wegovy pill is a once-a-day tablet of semaglutide 25 mg, made by Novo Nordisk.
It uses the same drug as injectable Wegovy and as Rybelsus (oral semaglutide for type 2 diabetes), but at a higher dose approved for weight loss.3 The U.S. FDA approved it on 22 December 2025.1
Foundayo is the brand name for orforglipron, a once-a-day tablet made by Eli Lilly. The U.S. FDA approved it on 1 April 2026.2
Both medications mimic the gut hormone GLP-1, which signals fullness, slows digestion, and quiets the constant intrusive thoughts about food that many people describe as ‘food noise’.
Where they differ is in how the drug is built, and how that affects the way you take it.
Peptide vs small molecule
Semaglutide is a peptide. Peptides are chains of amino acids and are broken down by stomach acid before they reach the bloodstream.
That’s why Wegovy was first developed as an injection.
To make a tablet, Novo Nordisk added an absorption enhancer called SNAC (salcaprozate sodium), which neutralises the acid around the tablet long enough for the semaglutide to pass through the stomach lining.6
SNAC only works when the stomach is empty and with no more than 120 ml of plain water alongside the tablet. Food, other drinks, and even other tablets reduce absorption.5
Orforglipron is a small synthetic compound rather than a peptide. It isn’t broken down by stomach acid, so it doesn’t need an absorption enhancer or a fasting window.4
The Wegovy pill is a peptide engineered to survive the stomach long enough to be absorbed; Foundayo is a non-peptide that the stomach doesn’t damage in the first place.
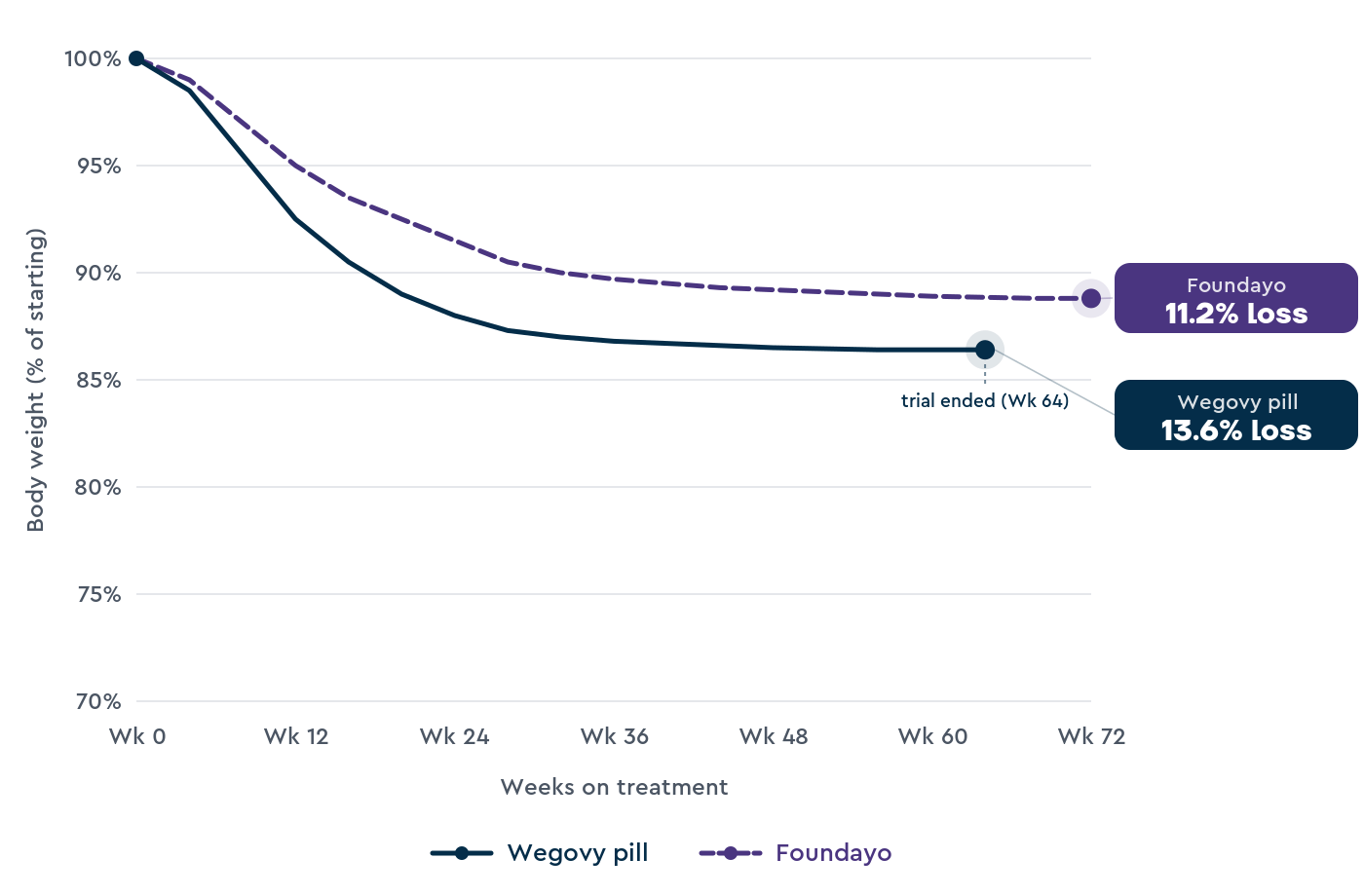
Average weight loss in trials
The two medications were tested in separate Phase 3 trials (large human trials with thousands of participants).
No trial has directly compared the two, so the comparison has to be drawn across studies with slightly different designs and populations.
OASIS 4 tested the Wegovy pill in 307 adults with obesity or overweight (with a weight-related condition), without type 2 diabetes.
Participants took 25 mg of oral semaglutide or a placebo daily for 64 weeks, alongside lifestyle changes.3
Results from OASIS 4:
- An average weight loss of 13.6% on oral semaglutide vs 2.2% on placebo
- 76.3% of people on the pill lost at least 5% of their body weight
- Around 30% lost 20% or more
- For people who completed the full 64 weeks, the average weight loss reached 16.6%
ATTAIN-1 tested Foundayo in 3,127 adults with obesity or overweight (with a weight-related medical problem), without type 2 diabetes.
Participants took orforglipron 6 mg, 12 mg, 36 mg, or a placebo daily for 72 weeks.4
Results from ATTAIN-1 at the highest dose (36 mg):
- An average weight loss of 11.2% on orforglipron vs 2.1% on placebo
- 54.6% lost at least 10% of their body weight
- 36% lost at least 15%
- 18.4% lost 20% or more
- For people who completed the full 72 weeks, the average weight loss reached 12.4%
The ATTAIN-1 trial used a 36 mg capsule.
The approved Foundayo product is a tablet, and the 17.2 mg tablet delivers the same dose as the 36 mg capsule.
Average weight loss at the approved 17.2 mg tablet dose is 11.1%, almost identical to the 11.2% reported with the 36 mg capsule in the trial.2
Both medications produce less weight loss than injectable Mounjaro (around 22.5% at the highest dose) and slightly less than injectable Wegovy (14.9% at 2.4 mg, or 20.7% at Wegovy 7.2 mg).3,9
Side effects compared
Side-effect profiles are similar. Both medications work on the same receptor (they mimic the same hormone), and the most common side effects are gastrointestinal and dose-related.
In OASIS 4, gastrointestinal side effects occurred in 74% of people on the Wegovy pill vs 42% on placebo.3 The most common were:
- Nausea (46.6% on the pill vs 18.6% on placebo)
- Vomiting (30.9% vs 5.9%)
- Diarrhoea
- Constipation
Stopping the medication due to side effects was 7% with oral semaglutide and 6% with placebo, which is unusually close for a GLP-1 trial, as the placebo group typically has fewer side effects.3
In ATTAIN-1, the most common side effects on Foundayo were nausea, diarrhoea, constipation, vomiting, indigestion, and abdominal pain.4 Most were mild to moderate and occurred during the dose-escalation phase.
Stopping the medication due to side effects in ATTAIN-1 ranged from 5.3% to 10.3% across the orforglipron dose groups, compared to 2.7% with placebo.4
Hair loss is listed on the Foundayo label, reported in 4 to 5% of people on the medication vs 2% on placebo, and more often in women (7%) than men (under 1%).2
Hair loss isn’t unique to Foundayo. It’s reported across the GLP-1 class (around 3% on injectable Wegovy, 5 to 6% on injectable Mounjaro) and is usually a temporary response to rapid weight loss rather than a direct effect of the drug.
Hair loss usually eases once weight stabilises. If you’re hair-loss conscious, mention it to your prescriber before starting either medication.
Both medications carry a boxed warning about thyroid C-cell tumours, based on findings in animal studies.
The risk hasn’t been confirmed in humans, but neither is recommended if you have a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2.
The fasting rule and what it means in practice
The Wegovy pill has to be taken on an empty stomach, first thing in the morning, with no more than 120 ml of plain water.
You can’t eat or drink anything else, or take any other oral medications, for at least 30 minutes after taking the tablet.5
If you take the pill with food or a normal-sized drink, less of the semaglutide is absorbed, and the medication won’t be as effective.
That routine is workable for many people, especially those already used to morning medications.
It’s a meaningful daily constraint for early-morning travel, irregular shifts, taking other tablets, or simply not wanting to wait half an hour for breakfast and coffee.
Foundayo has none of those rules. You can take the tablet whenever it fits your routine, with food, with coffee, with a full glass of water, or with other medications.2
Adherence to daily medications drops quickly when the routine is restrictive, so a pill that’s harder to take consistently can produce less weight loss in real-world use than a pill that can be taken under any conditions.
UK availability and likely order of arrival
Neither medication is approved in the UK yet.
Novo Nordisk submitted oral semaglutide 25 mg to the MHRA and the European Medicines Agency in the second half of 2025, and UK approval is expected in late 2026.1
Eli Lilly has confirmed Foundayo has been submitted in more than 40 countries, including the UK, but no MHRA timeline has been announced.
Given how recently the FDA approved Foundayo, UK approval is more likely towards the end of 206 or 2027.2
Private providers will offer the Wegovy pill first, with Foundayo arriving the following year. NHS access in either case would depend on a NICE health technology assessment after MHRA approval, which usually adds another 6 to 9 months.
UK pricing for either medication hasn’t been confirmed. In the U.S., both start at around $149 a month through self-pay channels.
Foundayo’s self-pay cost rises with the dose, reaching around $349 a month at 17.2 mg, while the Wegovy pill is priced at a single 25 mg dose.1,2
The only oral GLP-1 available in the UK is Rybelsus, licensed for type 2 diabetes only and at a lower dose (up to 14 mg) than Wegovy.
Injectable Wegovy and injectable Mounjaro are both available through private providers and, in some cases, the NHS.
Which oral GLP-1 might suit you
The trial numbers favour the Wegovy pill by a small margin for average weight loss, but the stricter dosing protocol may make Foundayo more appealing for some people.
If you can’t keep a morning fasting routine
Foundayo is the more flexible option, with no empty stomach, no water cap, and no 30-minute wait.
For shift workers, frequent travellers, or anyone who takes other morning medications, the lack of timing rules is the more useful feature.
If you want the strongest evidence behind your tablet
The Wegovy pill has slightly higher average weight loss in trials (13.6% vs 11.2%), and semaglutide has years of safety and outcome data behind it across injectable Wegovy, injectable Ozempic, and Rybelsus.3
If you’re switching from injectable Mounjaro or Wegovy
Both pills are options for maintaining weight loss after an injectable course.
In the ATTAIN-MAINTAIN trial, people who switched from injectable Wegovy to Foundayo regained an average of 1 kg over 52 weeks, while people who switched from injectable Mounjaro to Foundayo regained an average of 5 kg.7
Across both groups, participants maintained approximately 75-80% of their weight loss.
No equivalent trial has been published for the Wegovy pill, although it’s the same drug as injectable Wegovy at a different dose, making the switch a straightforward conversion in clinical practice.
Maintaining muscle mass on weight-loss medication
Both pills produce meaningful weight loss, and a portion of that weight loss can come from muscle and other lean tissues, such as bone, rather than fat.
The three things that help maintain muscle mass during weight loss apply to either medication: losing weight at a steady pace, eating around 1.2 to 1.6 grams of protein per kg of body weight per day (a serving the size of the palm of your hand at each meal), and adding resistance training a couple of times a week.
Frequently asked questions
Which is more effective for weight loss: Wegovy or Foundayo?
In separate trials, the Wegovy pill produced an average weight loss of 13.6% over 64 weeks, and Foundayo produced 11.2% over 72 weeks at the highest tested dose.34
The Wegovy pill seems to support more weight loss on average, but no trial has directly compared the two.
Which will be available in the UK first?
The Wegovy pill is expected to be approved by the MHRA in late 2026. Foundayo is expected to follow in 2027, although Eli Lilly hasn’t confirmed a UK timeline.12
Can you eat normally when taking Foundayo?
Yes. Foundayo can be taken at any time of day, with or without food, with no restrictions on what you drink.2
Why does the Wegovy pill have to be taken on an empty stomach?
Semaglutide is a peptide and would normally be broken down by stomach acid.
The pill includes an absorption enhancer (SNAC) that helps the drug cross the stomach lining, but it only works when the stomach is empty and with no more than 120 ml of water.6
Is the Wegovy pill the same as Rybelsus?
Both contain oral semaglutide, but they’re different products.
Rybelsus is licensed for type 2 diabetes at doses up to 14 mg. The Wegovy pill is a higher 25 mg dose approved for weight loss.
Can I take Foundayo if I’m on injectable Wegovy or Mounjaro?
Switching between GLP-1 medications needs to be discussed with your prescriber.
In the ATTAIN-MAINTAIN trial, people who switched from injectable Wegovy to Foundayo regained an average of 1 kg over 52 weeks.
People who switched from injectable Mounjaro to Foundayo regained around 5 kg, which is consistent with Mounjaro producing more weight loss to begin with.7
What are the side effects of each medication?
Both medications cause similar gastrointestinal side effects: nausea, vomiting, diarrhoea, and constipation.
These are usually mild to moderate and most common during the early weeks of treatment, while the dose is being increased.34
Foundayo also lists hair loss as a common side effect, although hair loss is reported across the GLP-1 class and is usually a temporary response to rapid weight loss.2
Is there a Mounjaro pill?
No. Tirzepatide, the drug in Mounjaro, hasn’t been developed as a tablet. Eli Lilly has instead developed Foundayo (orforglipron) as a separate oral GLP-1 medication.
Can I take other tablets at the same time as the Wegovy pill?
No. You need to wait at least 30 minutes after taking the Wegovy pill before taking any other oral medication.5 Foundayo has no such restriction.
If you’re considering an oral GLP-1 once it reaches the UK, our programme combines GLP-1 treatment with dietitian-led behaviour change support, so you build the eating and exercise habits that help you avoid weight regain in the future.
Second Nature’s published research found that members taking semaglutide alongside dietitian-led support achieved an average weight loss of 19.1% at 12 months, with 77.7% achieving at least 10% weight loss.8
Second Nature's Mounjaro and Wegovy programmes
Second Nature provides Mounjaro or Wegovy as part of our Mounjaro and Wegovy weight-loss programmes.
Why choose Second Nature over other medication providers, assuming you're eligible?
Because peace of mind matters.
We've had the privilege of working with the NHS for over eight years, helping people across the UK take meaningful steps toward a healthier, happier life.
Our programmes are designed to meet people where they are, whether that means support with weight loss through compassionate one-to-one health coaching, or access to the latest weight-loss medications (like Mounjaro and Wegovy) delivered alongside expert care from a multidisciplinary team of doctors, psychologists, dietitians, and personal trainers.
At the heart of everything we do is a simple belief: real, lasting change comes from building better habits, not relying on quick fixes. We're here to support that change every step of the way.
With over a decade of experience, thousands of lives changed, and a long-standing record of delivering programmes used by the NHS, we believe we're the UK's most trusted weight-loss programme.
We hope to offer you something invaluable: peace of mind, and the support you need to take that first step.
Take home message
The Wegovy pill and Foundayo are the two oral GLP-1 weight-loss medications expected to reach the UK in the next 12 to 24 months.
The Wegovy pill produces slightly more weight loss in trials (13.6% vs 11.2%) and is likely to arrive first, in late 2026.
Foundayo is easier to take day to day, with no fasting, water, or timing restrictions, and is expected to follow in 2027.
For people who can’t keep a strict morning fasting routine, Foundayo’s flexibility is likely to be more appealing.
For people who want the strongest trial evidence and the earliest UK access, the Wegovy pill might be the preferred choice.
Both are less effective than injectable Mounjaro and Wegovy, which remain the most effective GLP-1 medications for weight loss in the UK.
References
-
- Novo Nordisk. (2025). Wegovy pill approved in the US as first oral GLP-1 for weight management. Press release, 22 December 2025.
- Eli Lilly. (2026). FDA approves Lilly’s Foundayo (orforglipron), the only GLP-1 pill for weight loss that can be taken any time of day without food or water restrictions. Press release, 1 April 2026.
- Wharton, S., et al. (2025). Oral semaglutide at a dose of 25 mg in adults with overweight or obesity. New England Journal of Medicine, 393(11). PMID: 40934115.
- Wharton, S., et al. (2025). Orforglipron, an oral small-molecule GLP-1 receptor agonist, for obesity treatment. New England Journal of Medicine, 393(18). PMID: 40960239.
- U.S. Food and Drug Administration. (2025). Wegovy (oral semaglutide) prescribing information.
- Buckley, S.T., et al. (2018). Transcellular stomach absorption of a derivatized glucagon-like peptide-1 receptor agonist. Science Translational Medicine, 10(467).
- Aronne, L.J., et al. (2026). Orforglipron for maintenance of body weight reduction: the double-blind, randomized phase 3b ATTAIN-MAINTAIN trial. Nature Medicine. DOI: 10.1038/s41591-026-04386-7.
- Richards, R., et al. (2025). A Remotely Delivered GLP-1RA-Supported Specialist Weight Management Program in Adults Living With Obesity: Retrospective Service Evaluation. JMIR Formative Research, 9(1), e72577.
- Wharton, S., et al. (2025). Once-weekly semaglutide 7.2 mg in adults with obesity (STEP UP): a randomised, controlled, phase 3b trial. The Lancet Diabetes & Endocrinology, 13(11), 949-963. PMID: 40961952.