How much weight do people lose on each
The figures most often quoted side by side come from two different trials, in different patient groups, over different time periods.
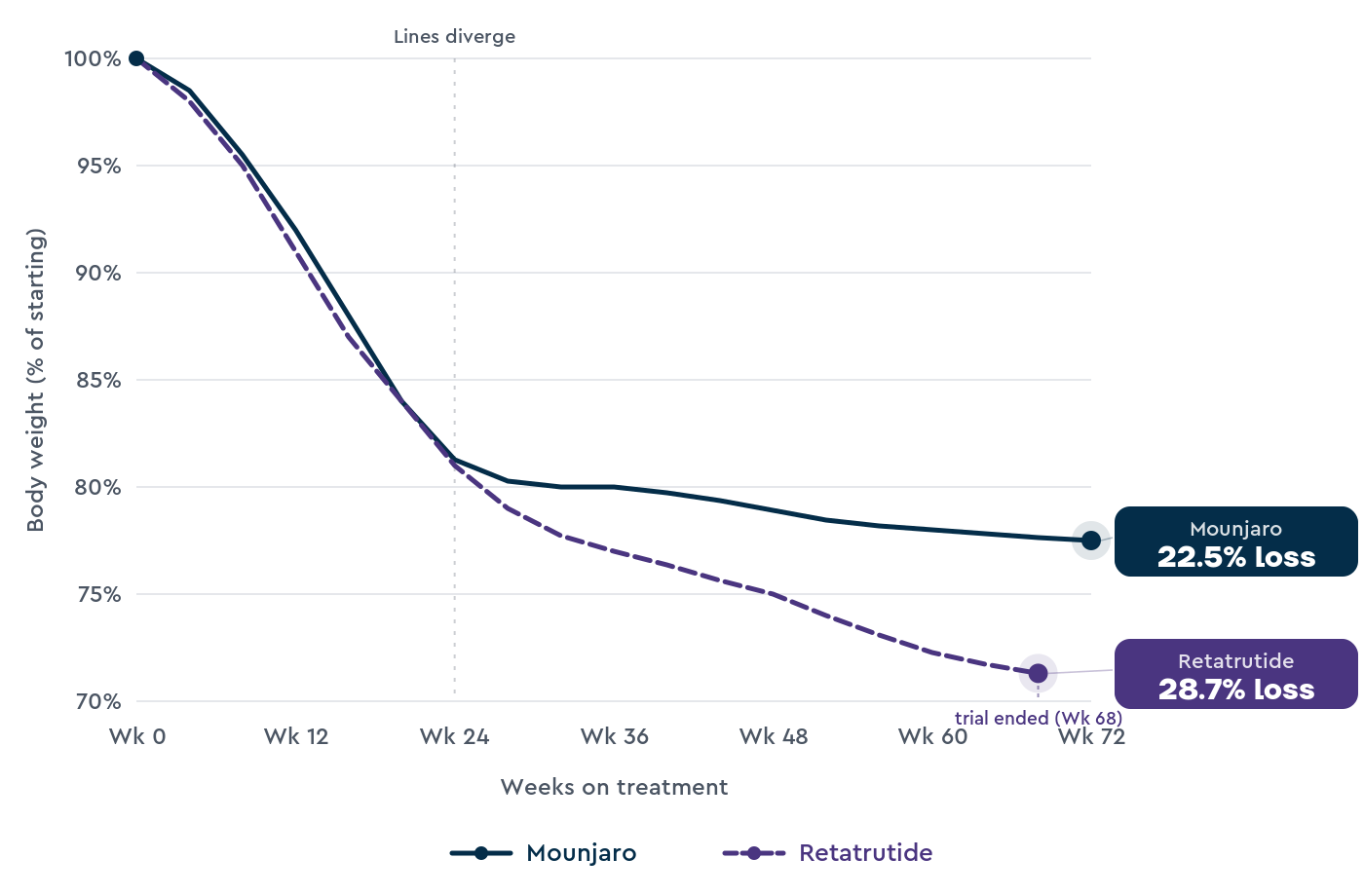
For Mounjaro, SURMOUNT-1 enrolled 2,539 adults living with obesity (and weight-related health problems, but not type 2 diabetes) and followed them for 72 weeks.1
Average weight loss was 22.5% at 15 mg, 21.4% at 10 mg, and 16.0% at 5 mg, compared with 3.1% on placebo.
For retatrutide, TRIUMPH-4 enrolled 445 adults living with obesity and knee osteoarthritis, and followed them for 68 weeks.2
Average weight loss was 28.7% at 12 mg, 26.4% at 9 mg, and 2.1% on placebo. 58.6% of people on the 12 mg dose lost at least 25% of their body weight.
TRIUMPH-4 was shorter than SURMOUNT-1, smaller, and the population had knee osteoarthritis, which can affect how easily people stay active.
The full TRIUMPH-4 paper hasn’t yet been published in a peer-reviewed journal; the figures above come from Lilly’s December 2025 press release.2
The 28.7% figure is the largest average weight loss reported in any Phase 3 obesity trial so far.
Earlier evidence for retatrutide comes from a Phase 2 trial in NEJM in 2023, which reported 24.2% weight loss at 12 mg over 48 weeks.3 TRIUMPH-4 confirms and extends that earlier finding.
Retatrutide’s main obesity trial, TRIUMPH-1, is ongoing in adults living with obesity but without knee osteoarthritis. Published results are expected later in 2026.
Retatrutide looks stronger in the trial data so far, but no trial has directly compared the two medications.
We need to be cautious when comparing results from different trials, as the design can inherently influence outcomes.
| Measure |
Mounjaro (tirzepatide) |
Retatrutide |
| How it works |
Works on 2 hormone receptors (GLP-1 and GIP) |
Works on 3 hormone receptors (GLP-1, GIP, and glucagon) |
| Largest average trial weight loss |
22.5% at 15 mg over 72 weeks (SURMOUNT-1, n=2,539)1 |
28.7% at 12 mg over 68 weeks (TRIUMPH-4, n=445, obesity with knee osteoarthritis)2 |
| Maintenance doses |
5, 10, or 15 mg once a week |
4, 9, or 12 mg once a week (in trials) |
| Administration |
Subcutaneous injection, once a week |
Subcutaneous injection, once a week |
| UK licence |
MHRA-approved for managing obesity, November 2023; NICE TA1026 December 20245 |
Not approved. FDA submission expected late 2026; MHRA approval realistically late 2027 to mid 2028 |
| UK availability (May 2026) |
NHS (specialist services, expanding) or private prescription |
Not legally available |
| Cardiovascular outcomes data |
SURMOUNT-MMO trial ongoing |
No completed outcomes trial yet |
How retatrutide and Mounjaro work differently
Both medications work by mimicking hormones that influence our hunger and appetite, but retatrutide mimics one more hormone than Mounjaro.
The triple-action of retatrutide is likely why it seems to support more weight loss than Mounjaro.
Mounjaro is a double-action drug that mimics two hormones, GLP-1 and GIP.
GLP-1 helps regulate appetite and blood sugar, slows the rate at which our stomach empties, and helps our pancreas release insulin after eating.
GIP works alongside GLP-1 to regulate insulin response and may also affect fat storage.
Retatrutide has a triple-action. It mimics the same two hormones as Mounjaro, plus a third hormone, glucagon.3
Glucagon is the hormone most people associate with raising blood sugar, but it has a second function. It increases our basal metabolic rate (how much energy we burn at rest), meaning we burn more energy throughout the day, and it promotes the breakdown of stored fat for fuel.
The triple action means retatrutide both reduces appetite (like Mounjaro) and increases how much energy our body burns even when we aren’t moving.
By also mimicking glucagon, retatrutide may increase the likelihood of experiencing side effects compared to Mounjaro.
Side effects compared
Both medications share similar side effects affecting the stomach and gut because they both mimic GLP-1. Retatrutide adds a few effects that come from the glucagon component.
For Mounjaro, the most common side effects are nausea, diarrhoea, constipation, vomiting, and reduced appetite.1
Most are mild to moderate and resolve with the slow titration schedule (starting at 2.5 mg, increasing every 4 weeks).
In SURMOUNT-1, around 4 to 7% of people on tirzepatide stopped the medication because of side effects, compared with 2.6% on placebo.1
Mounjaro has been prescribed in the UK since November 2023, and a real-world safety profile is now established.
For retatrutide, the stomach and gut profile is similar but more frequent at the highest dose. In TRIUMPH-4, nausea occurred in 43% of people on 12 mg, vomiting in 21%, and diarrhoea in 33%.2
Around 11% of people on the highest dose stopped retatrutide because of side effects, roughly double the rate seen on Mounjaro in SURMOUNT-1.2
Retatrutide also has two specific concerns that aren’t as common on Mounjaro.
The first is heart rate. Across the Phase 2 and Phase 3 trials, heart rate increased by approximately 6.7 bpm at the 12 mg dose, peaking around week 24 and declining thereafter.3
For comparison, Mounjaro and Wegovy typically raise resting heart rate by 2 to 4 bpm. Retatrutide’s increase is roughly two to three times larger.
The second is dysesthesia, an abnormal skin sensation often described as burning or tingling.
In TRIUMPH-4, this was reported in 20.9% of people on 12 mg, 8.8% on 9 mg, and 0.7% on placebo.2
These events didn’t lead to people stopping the medication, but regulators are watching it closely across the wider Phase 3 programme.
Irregular heartbeats are the third area regulators are watching. Across the retatrutide trials so far, rates have ranged from 2 to 11% in active groups (depending on dose) versus 2% on placebo.
None have been classified as serious, and there’s no clear pattern of increased major cardiac events.
But retatrutide hasn’t yet completed a dedicated cardiovascular outcomes trial, so it’s unclear what effect it will have on heart health in the long term.
Mounjaro doesn’t have the same heart rate or dysesthesia concerns, and its longer time on the UK market means we have a clearer view of who tolerates it well.
What happens if you stop taking the medication
Both medications work for as long as you take them. When the medication stops, the appetite-reducing effect fades, and most people gain weight back unless they have other support in place.
The clearest evidence comes from SURMOUNT-4, where adults who had lost an average of 20.9% on Mounjaro over 36 weeks were then either continued on the drug or switched to placebo for another year.4
The people who continued on Mounjaro lost a further 5.5%. The people who switched to placebo regained 14% of their starting weight, retaining only about a third of the weight loss they’d initially achieved.4
Still, this research shows what happens when you stop the medications suddenly without a gradual decrease in dose (a taper), and with adequate lifestyle support alongside it.
Emerging research suggests that people who come off the medication gradually, with structured habit-change support, are more likely to maintain their weight loss.
Cost and access in the UK
Mounjaro is available in the UK through three routes.
The first is the NHS, where it’s recommended by NICE (technology appraisal TA1026, December 2024) for adults with a BMI of at least 35 (or 32.5 for some ethnicities) who have at least three weight-related health problems.5
NHS access is rolling out in phases through specialist weight-management services and primary care.
The second is private prescription, where Second Nature’s Mounjaro programme and other UK providers offer it through a clinical assessment.
The third is online pharmacies, which can prescribe it after a clinical review.
Retatrutide isn’t legally available in the UK. As of May 2026, no UK doctor can prescribe it on the NHS or privately.
Eli Lilly is targeting an FDA submission in late 2026, with an MHRA submission expected to follow within a few months.
A realistic UK approval window is late 2027 to mid 2028, with NHS availability through NICE technology appraisal arriving in 2029 at the earliest.
Some online sellers advertise retatrutide as a research chemical or compounded peptide.
These are illegal in the UK and have been the focus of MHRA enforcement action against UK facilities producing counterfeit GLP-1 medicines.
Counterfeit pens have contained insulin, saline, or unidentified substances. Buying retatrutide online is unsafe and we don’t recommend it.
We’ve covered the details in our guide on buying retatrutide online safely in the UK.
UK private prices for Mounjaro currently sit in a range of around £150 to £350 a month, depending on dose and provider, with higher doses sitting at the top of the range.
Retatrutide’s UK price won’t be known until MHRA approval and NICE appraisal are complete, but it’s likely to sit in a similar range to current GLP-1 medications, with NHS pricing typically lower than private list prices.
Cardiovascular and long-term safety
Mounjaro has more long-term safety data because it’s been prescribed for type 2 diabetes in the U.S. since 2022 and for managing obesity in the UK since November 2023.
Its dedicated cardiovascular outcomes trial, SURMOUNT-MMO, is ongoing.
Trials of tirzepatide in obesity and type 2 diabetes have shown improvements in blood pressure, cholesterol, and HbA1c (a marker of blood sugar control) alongside weight loss, though the SURMOUNT-MMO data will be the clearest evidence on cardiovascular outcomes.
Retatrutide doesn’t yet have a completed cardiovascular outcomes trial.
The TRIUMPH programme is collecting cardiovascular data alongside its weight-loss measures, and a dedicated outcomes trial is expected once retatrutide is approved.
The increase in heart rate in retatrutide trials is the side effect that regulators and clinicians are watching closely.
Beyond cardiovascular data, retatrutide is also being studied in adults living with type 2 diabetes (TRIUMPH-2) and in metabolic dysfunction-associated steatohepatitis (MASH), a form of advanced fatty liver disease.
Early results across these areas are promising, but full data haven’t yet been published.
Muscle mass and body composition
One of the concerns around GLP-1 medications has been the risk of losing muscle and other lean tissues alongside fat.
In the published Mounjaro trials, roughly 25-30% of total weight loss is from lean mass, including muscle.
The same range applies to most GLP-1 medications. Retatrutide’s body composition data are broadly similar in the Phase 2 results, with the higher total weight loss accompanied by a greater absolute reduction in lean mass.
Muscle helps the body regulate blood sugar, supports strength for everyday tasks, and is a strong predictor of balance and independence as we get older.
There are three things to focus on to help maintain muscle mass during weight loss
- Lose weight slowly at a rate of around 1lb per week
- Eat enough protein, roughly a portion the size of the palm of your hand in each meal
- Try to do some resistance training
Switching considerations
Switching from Mounjaro to retatrutide isn’t a real option in 2026, because retatrutide isn’t approved yet.
The question still comes up because Reddit communities have reported people switching after a Mounjaro plateau (using illegally sourced product) and starting to lose weight again.
We can’t comment on those self-reports because the products people are using haven’t been verified for purity, dose accuracy, or sterility.
If retatrutide is approved in the UK and you’re already taking Mounjaro, switching would normally involve stopping Mounjaro, allowing a washout period of one week, and starting retatrutide at an equivalent dose.
A clinician would assess whether switching is right for you, based on how Mounjaro is working, your tolerance, and your medical history.
For now, if Mounjaro isn’t working well for you, the practical alternatives are a dose adjustment, switching to Wegovy, or focusing more on the lifestyle and behaviour-change side of the programme.
We’ve covered stopping Mounjaro in our guide on coming off Mounjaro.
Who should consider which
In May 2026, there’s only one real choice between these two medications, because retatrutide isn’t legally available in the UK.
The realistic answer is Mounjaro, if it’s appropriate for you and you’ve spoken with a healthcare professional.
Mounjaro suits adults who meet the NICE eligibility criteria (BMI of at least 35, or 32.5 for some ethnicities, with weight-related health problems), and adults using private services who have a BMI of at least 30, or 27.5 with health complications.5
It’s a useful option for people who haven’t responded enough to lifestyle changes alone.
Retatrutide will be a real option from late 2027 at the earliest, once it’s approved.
Based on the trial profile, it may suit people who haven’t lost enough weight on GLP-1-based medications, or who need the largest possible weight loss for severe obesity-related conditions.
The heart-rate increase means it might not be the right choice for people with significant heart-rhythm conditions, but that’s a clinical judgement to make once full data is available.
Wegovy (semaglutide) is the third option in this category and is also approved in the UK.
In SURMOUNT-5, which directly compared the two medications, Mounjaro 15 mg produced 20.2% weight loss, while Wegovy 2.4 mg produced 13.7% over 72 weeks.6
A higher 7.2 mg dose of Wegovy has since been approved by the MHRA (January 2026). In the STEP-UP trial, the 7.2 mg dose produced 20.7% weight loss at 72 weeks, much closer to Mounjaro 15 mg than the 2.4 mg dose tested in SURMOUNT-5.
Mounjaro tends to produce more weight loss; Wegovy has more cardiovascular outcomes data from the SELECT trial, which is relevant if you have established cardiovascular disease.
Frequently asked questions
Is retatrutide stronger than Mounjaro?
In the trial data published so far, retatrutide has produced larger average weight loss than Mounjaro, with 28.7% at 68 weeks in TRIUMPH-4 versus 22.5% at 72 weeks in SURMOUNT-1.1,2
But these are different trials in different patient groups over different time periods. No trial has directly compared the two medications.
Should I wait for retatrutide before starting Mounjaro?
For most people, no. Retatrutide is at least 18 months away from MHRA approval, and probably longer before it’s available on the NHS.
Mounjaro produces clinically significant weight loss for most people who tolerate it, and starting now means you spend less time at a higher weight, which compounds health benefits over time.
If retatrutide is later approved and suits your situation, switching is possible.
When will retatrutide be available in the UK?
Eli Lilly is targeting an FDA submission in late 2026. MHRA submission usually follows within a few months.
A realistic UK approval window is late 2027 to mid 2028. NHS availability through NICE technology appraisal would arrive in 2029 at the earliest.
Can I get retatrutide on the NHS?
No. Retatrutide isn’t approved by the MHRA, hasn’t been appraised by NICE, and isn’t on the NHS formulary.
None of those steps will be complete before late 2027 at the earliest. We’ve covered the full UK timeline in our retatrutide and the NHS guide.
Is it safe to buy retatrutide online?
No. Retatrutide isn’t legally sold in the UK in any form.
The MHRA has taken enforcement action against UK facilities producing counterfeit GLP-1 medicines, including retatrutide.
Counterfeit pens have contained insulin, saline, or unidentified substances.
Our retatrutide sourcing safety guide covers why purchasing the medication on the grey market isn’t worth the risk.
What’s the difference between a triple agonist and a dual agonist?
Mounjaro is a dual agonist: it works on two hormone receptors (GLP-1 and GIP).
Retatrutide is a triple agonist: it works on three (GLP-1, GIP, and glucagon).3
The third receptor (glucagon) increases the amount of energy our body burns at rest, which is the main reason trial weight loss has been higher.
Does retatrutide raise heart rate more than Mounjaro?
Yes. Retatrutide’s heart-rate increase has averaged around 6.7 bpm at 12 mg, peaking around week 24 and declining thereafter.3
Mounjaro and Wegovy typically raise resting heart rate by 2 to 4 bpm, so retatrutide’s increase is roughly two to three times larger.
The planned cardiovascular outcomes trial will help answer whether this translates to any longer-term risk.
What happens if I stop taking these medications?
Most people regain a significant portion of the weight they lost.
In SURMOUNT-4, adults who had lost an average of 20.9% on Mounjaro then switched to placebo regained 14% of their starting weight over the following year, retaining only about a third of the weight loss they’d initially achieved.4
Retatrutide’s withdrawal data isn’t published yet, but the same pattern is expected.
If retatrutide is stronger, why should I take Mounjaro?
Retatrutide isn’t legally available in the UK, so it isn’t a current option.
Even after retatrutide is approved, Mounjaro will still suit many people because it has a longer real-world safety record, a clearer cardiovascular profile, and produces clinically significant weight loss for most people who tolerate it.
Can I switch from Mounjaro to retatrutide later?
Once retatrutide is approved in the UK, a switch would normally involve stopping Mounjaro, a washout of around four to six weeks, and starting retatrutide at the lowest dose with slow titration.
A clinician would assess whether switching is right for you, based on how Mounjaro is working, your tolerance, and your medical history.
How does Mounjaro compare to Wegovy?
In the SURMOUNT-5 trial, which directly compared the two medications, Mounjaro 15 mg produced 20.2% weight loss, while Wegovy 2.4 mg produced 13.7% over 72 weeks.6
A higher dose of 7.2 mg of Wegovy has since been approved by the MHRA (January 2026), leading to ~20% weight loss.
Mounjaro tends to produce more weight loss; Wegovy has more cardiovascular outcomes data.
Take home message
Retatrutide and Mounjaro are both injectable obesity medications at different stages of development.
Mounjaro is approved, available, and backed by years of clinical data.
Retatrutide has produced the largest weight loss reported in any Phase 3 obesity trial so far, but a UK launch is at least 18 months away.
The extra hormone that retatrutide mimics, glucagon, helps explain why the trial numbers are higher. It also brings side effects we don’t see with Mounjaro, including a larger increase in heart rate.
If you’re choosing a weight-loss medication today, the practical choice is between Mounjaro and Wegovy.
Second Nature’s medication programme combines Mounjaro with structured support from registered dietitians, including the balanced plate model (half vegetables, quarter protein, quarter complex carbohydrates, plus a serving of fat) and habit-building tools.
Our peer-reviewed JMIR study of the GLP-1RA-supported programme reported an average weight loss of 19.1% at 12 months among active subscribers, with 77.7% achieving at least 10% weight loss.7
Second Nature's Mounjaro and Wegovy programmes
Second Nature provides Mounjaro or Wegovy as part of our Mounjaro and Wegovy weight-loss programmes.
Why choose Second Nature over other medication providers, assuming you're eligible?
Because peace of mind matters.
We've had the privilege of working with the NHS for over eight years, helping people across the UK take meaningful steps toward a healthier, happier life.
Our programmes are designed to meet people where they are, whether that means support with weight loss through compassionate one-to-one health coaching, or access to the latest weight-loss medications (like Mounjaro and Wegovy) delivered alongside expert care from a multidisciplinary team of doctors, psychologists, dietitians, and personal trainers.
At the heart of everything we do is a simple belief: real, lasting change comes from building better habits, not relying on quick fixes. We're here to support that change every step of the way.
With over a decade of experience, thousands of lives changed, and a long-standing record of delivering programmes used by the NHS, we believe we're the UK's most trusted weight-loss programme.
We hope to offer you something invaluable: peace of mind, and the support you need to take that first step.
References
- Jastreboff, A.M. et al. (2022). Tirzepatide once weekly for the treatment of obesity (SURMOUNT-1). New England Journal of Medicine, 387(3), 205-216.
- Eli Lilly and Company. (2025, December 11). Lilly’s triple agonist, retatrutide, delivered weight loss of up to an average of 71.2 lbs along with substantial relief from osteoarthritis pain in first successful Phase 3 trial (TRIUMPH-4 press release).
- Jastreboff, A.M. et al. (2023). Triple-hormone-receptor agonist retatrutide for obesity: a Phase 2 trial. New England Journal of Medicine, 389(6), 514-526.
- Aronne, L.J. et al. (2024). Continued treatment with tirzepatide for maintenance of weight reduction in adults with obesity: the SURMOUNT-4 randomized clinical trial. JAMA, 331(1), 38-48.
- National Institute for Health and Care Excellence. (2024, December 23). Tirzepatide for managing overweight and obesity (TA1026).
- Aronne, L.J. et al. (2025). Tirzepatide as compared with semaglutide for the treatment of obesity (SURMOUNT-5). New England Journal of Medicine.
- Richards, R., Whitman, M., Wren, G., Campion, P. (2025). A Remotely Delivered GLP-1RA-Supported Specialist Weight Management Program in Adults Living With Obesity: Retrospective Service Evaluation. JMIR Formative Research, 9(1), e72577.