Weight-loss injections: comparison at a glance
| Feature |
Mounjaro (15 mg) |
Wegovy (7.2 mg) |
Wegovy (2.4 mg) |
Saxenda |
| Drug |
Tirzepatide |
Semaglutide |
Semaglutide |
Liraglutide |
| Average weight loss |
22.5% |
20.7% |
15–17% |
5–7% |
| Injection frequency |
Weekly |
Weekly (3 × 2.4 mg) |
Weekly |
Daily |
| NHS availability |
Limited (phased rollout from June 2025) |
Not yet (NICE review pending) |
Limited specialist services |
Tier 3 only (rarely used now) |
| Private cost/month |
~£300–375 |
~£250–300 (estimated) |
~£150–250 |
~£150–300 |
| Time to see results |
4–8 weeks |
4–8 weeks |
4–8 weeks |
8–12 weeks |
| Main side effects |
Nausea, diarrhoea |
Nausea, diarrhoea, dysaesthesia |
Nausea, constipation |
Nausea, headaches |
In their main trials, 57% of people lost 20% or more of their body weight on Mounjaro 15 mg, and around one in three lost 25% or more on Wegovy 7.2 mg.1,2
1. Tirzepatide (Mounjaro)
Tirzepatide, marketed as Mounjaro in the UK, remains the most effective weight-loss injection based on clinical trial data.
Mounjaro also became available on the NHS for weight management in June 2025, though access remains highly restricted and is reserved for individuals with the greatest clinical need.
How Mounjaro works
Mounjaro mimics two hormones: GLP-1 and GIP. This dual action reduces appetite and slows gastric emptying (the rate at which food leaves your stomach) more effectively than medications that mimic GLP-1 alone.1
The medication starts at 2.5 mg per week, increasing gradually to a maintenance dose of 5 mg, 10 mg, or 15 mg, depending on individual response and tolerability.
Clinical evidence
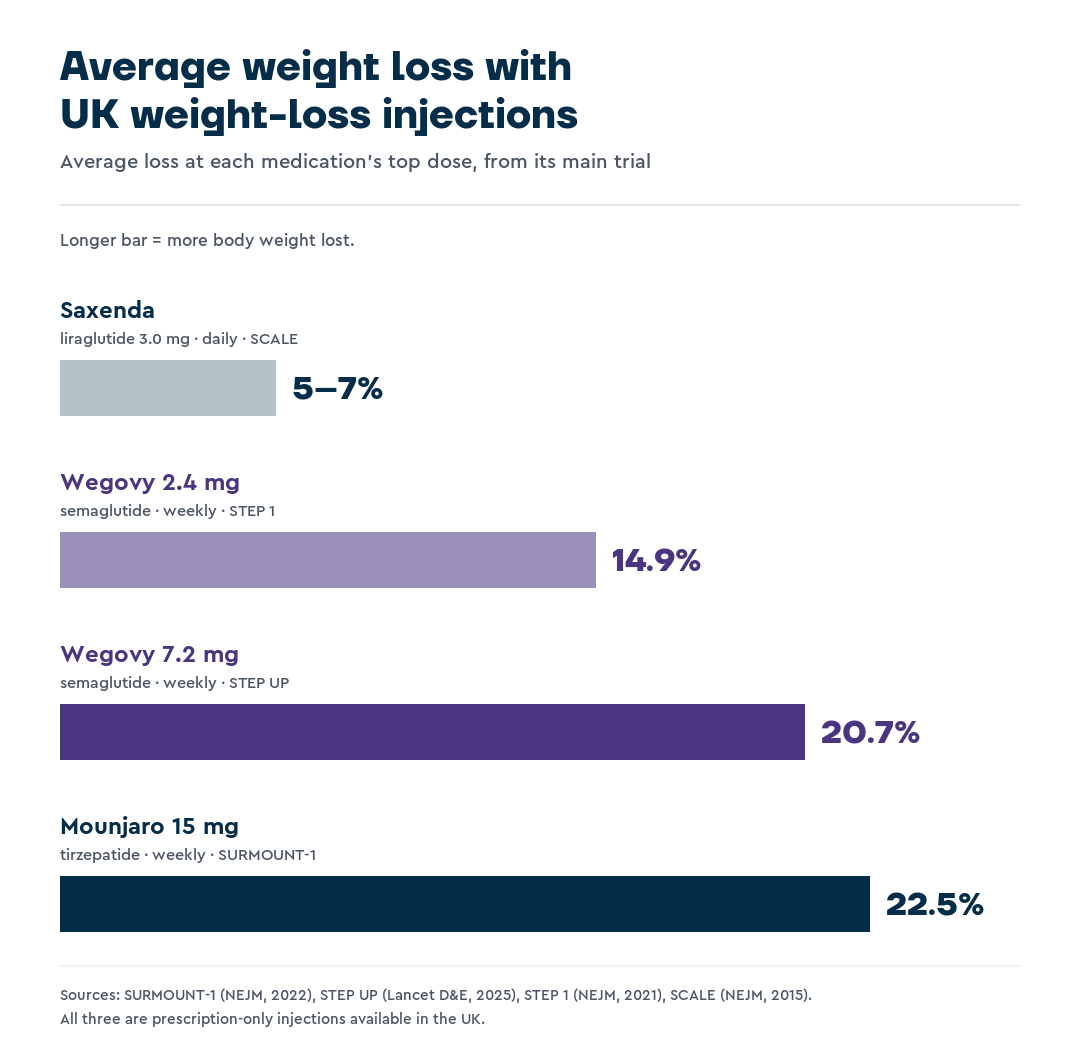
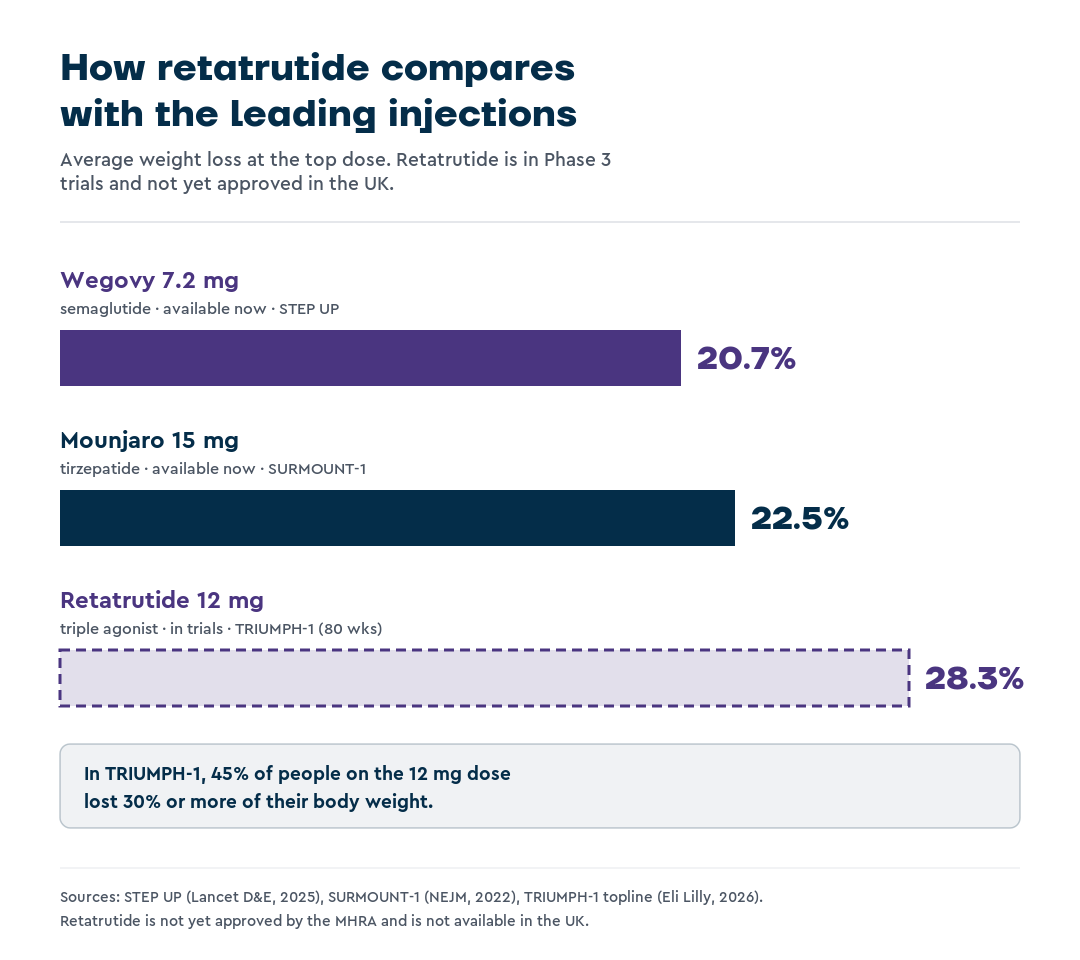
The SURMOUNT-1 trial found that participants without diabetes lost an average of 22.5% of body weight at the 15 mg dose after 72 weeks.
In the same trial, 91% lost at least 5% of their body weight, and 57% lost 20% or more.1
A 2025 trial published in the New England Journal of Medicine, which directly compared the two medications, found that tirzepatide produced greater weight loss than semaglutide 2.4 mg.3
Follow-up data from the SURMOUNT-4 trial shows the weight loss is maintained at 88 weeks with continued use.4
Who can take Mounjaro?
NICE approved Mounjaro for weight management (TA1026) in adults with a BMI of 35 or above with weight-related health conditions, or a BMI of 30 or above with specific comorbidities.5
You can’t take Mounjaro if you have a personal or family history of medullary thyroid cancer or Multiple Endocrine Neoplasia syndrome type 2.
People with a history of pancreatitis or severe gastrointestinal disease need careful assessment before starting.
The MHRA issued a Drug Safety Update in January 2025 about monitoring and managing gastrointestinal side effects with tirzepatide.6
The MHRA has also warned that Mounjaro may reduce the effectiveness of the combined oral contraceptive pill.
If you take the pill, use an additional barrier method such as condoms while on Mounjaro, or consider switching to a non-oral contraceptive like patches or an IUD.
The September 2025 price increase
Eli Lilly raised Mounjaro’s UK list price from September 2025 to bring it closer to global pricing levels.
The highest dose (15 mg) saw the steepest increase, with list prices rising from £122 to £330 per four-week pen.
In practice, private prescription costs typically range from £149 for the initial 2.5 mg dose to £300–375 for the 15 mg maintenance dose, depending on the provider.
This is a large increase from the pre-September prices of £92–122. Our guide to why Mounjaro’s price increased breaks down the new prices by dose.
This price increase has led many people to reconsider Wegovy as an alternative, particularly now that the higher 7.2 mg dose is available with comparable weight-loss results.
2. Semaglutide (Wegovy)
Wegovy has been one of the most widely prescribed weight-loss injections worldwide since its UK launch.
In January 2026, the MHRA approved a higher dose of 7.2 mg, which supports similar weight loss to Mounjaro.
Wegovy 7.2 mg
The STEP UP trial found that patients on Wegovy 7.2 mg lost an average of 20.7% of their body weight after 72 weeks, compared with 17.5% on the standard 2.4 mg dose.
A third of participants lost 25% or more of their body weight.2
This higher dose is not a starting dose. It’s intended for people who have already reached and tolerated the 2.4 mg dose but need additional support to continue losing weight or have plateaued.
Currently, Wegovy 7.2 mg is given as three consecutive 2.4 mg injections on the same day, using the existing 2.4 mg pen. A dedicated single-injection 7.2 mg pen is expected later in 2026.
How Wegovy differs from Ozempic
Both contain the drug semaglutide, but Wegovy uses higher doses specifically approved for weight loss. Ozempic’s maximum dose is 2 mg for type 2 diabetes.
Ozempic isn’t licensed for weight loss in the UK and shouldn’t be used off-label for this purpose.
Wegovy follows a 16-week dose escalation schedule, starting at 0.25 mg and increasing gradually to the maintenance dose.
Clinical evidence
The STEP 1 trial showed participants achieved an average 14.9% weight loss over 68 weeks with semaglutide 2.4 mg.7
In a real-world UK setting, Second Nature’s published research in JMIR Formative Research found that participants achieved an average weight loss of 19.1% at 12 months on a semaglutide-supported programme, with 77.7% achieving at least 10% weight loss.8
The SELECT trial demonstrated that semaglutide reduces major adverse cardiovascular events by 20% in people with existing heart disease and obesity.9
Three reasons people are choosing Wegovy in 2026
Wegovy now appeals to many people for three practical reasons.
The first is cost. Following Mounjaro’s price increase, Wegovy 2.4 mg typically costs £150 to £250 a month, less than Mounjaro at its maintenance doses of around £300 to £375 a month for the 15 mg dose. Wegovy 7.2 mg costs between £250 and £375 per month.
The second is cardiovascular disease evidence. Wegovy is currently the only weight-loss injection with heart-health benefits recognised on its label, based on the SELECT trial.
Mounjaro has published trials suggesting cardiovascular benefits, but these haven’t yet been added to its licence.
The third is its safety record. Wegovy has the longest real-world safety data of the newer weekly injections, with millions of doses given worldwide since 2021.
3. Liraglutide (Saxenda)
Saxenda is the Novo Nordisk brand of liraglutide 3.0 mg, a daily injection for weight management.
It’s now prescribed far less often than it was. The weekly injections produce greater average weight loss, and branded Saxenda has had long-term supply problems in the UK.
Since the patent expired in 2025, the same medication is increasingly supplied as generic liraglutide from other manufacturers.
Biocon’s version, Biolide, received MHRA approval in early 2025, and other generic brands such as Nevolat are also available privately.
If you’re offered liraglutide today, it’s likely to be one of these generic versions rather than branded Saxenda.
Clinical effectiveness
The SCALE trials demonstrated average weight loss of 5–7% over 56 weeks, which is modest compared with Mounjaro and Wegovy.10
63% of participants achieved at least 5% weight loss, meeting the threshold for clinically meaningful health improvements.
When Saxenda might still be appropriate
Saxenda may be considered if you have not previously tolerated weekly GLP-1 medications, prefer daily dosing (the ability to stop quickly if side effects occur), or cost is a primary concern, and Wegovy or Mounjaro are unaffordable.
On the NHS, liraglutide is still listed for specialist Tier 3 weight management services under NICE guidance (TA664), but it’s now rarely the first choice, having been largely replaced by Wegovy and Mounjaro.11
The daily injection schedule is also harder to maintain over the long term, with more people stopping the medication due to side effects than Wegovy or Mounjaro.
NHS vs private access
NHS access in 2026
NHS access to weight-loss injections has expanded since 2025, but remains limited for most people.
Mounjaro (tirzepatide): NICE approved tirzepatide for weight management in early 2025 (TA1026), and NHS prescribing through primary care began in June 2025 as part of a phased 12-year rollout.5
Our guide to getting Mounjaro on the NHS covers the pathway and what to ask your GP.
Initial eligibility is restricted to people with a BMI of 40 or above (adjusted for ethnicity) and at least four weight-related health conditions.
These include type 2 diabetes, high blood pressure, cardiovascular disease, and obstructive sleep apnoea. This threshold will gradually widen over the coming years, and availability varies by region and GP surgery.
Wegovy (semaglutide 2.4 mg): Available through NHS specialist Tier 3 weight management services, with eligibility typically requiring a BMI of 35 or above with health conditions. Waiting times range from 6 to 24 months, depending on location.
Wegovy 7.2 mg: Not yet available on the NHS. NICE will need to complete a cost-effectiveness review before NHS coverage is confirmed, likely towards late 2026.
Saxenda (liraglutide): Still covered for specialist Tier 3 services under NICE guidance (TA664), but rarely used now.
Branded Saxenda is in short supply, so any liraglutide prescribed today, whether on the NHS or privately, is increasingly a generic version such as Biolide or Nevolat.11
Private prescriptions
Private prescriptions remain the primary route for most people. Eligibility thresholds are lower than NHS criteria, typically requiring a BMI of 30 or above, or 27 with weight-related health conditions.
In January 2026, the General Pharmaceutical Council published updated inspection guidance covering weight-loss medications, online pharmacies, and prescribing standards.
Reputable providers must now conduct more thorough assessments than those provided by a simple online questionnaire.
When choosing a private provider, check for CQC registration, qualified prescribers who conduct proper medical assessments, and ongoing monitoring throughout treatment.
Insurance
Most UK private health insurance excludes weight-loss medications.
Side effects comparison
Common side effects across all GLP-1 medications
Gastrointestinal side effects are the most common, particularly in the first few weeks and during dose increases. For most people, these improve as the body adjusts.
Nausea affects 30 to 44% of users initially, and constipation or diarrhoea may alternate.
Fatigue and headaches occur in 10 to 15% of users, often related to reduced calorie intake rather than the medication itself.
Medication-specific patterns
| Side effect |
Mounjaro |
Wegovy 7.2 mg |
Wegovy 2.4 mg |
Saxenda |
| Nausea |
30% |
Not separately reported |
44% |
40% |
| Diarrhoea |
21% |
Not separately reported |
30% |
21% |
| Constipation |
17% |
Not separately reported |
24% |
19% |
| GI events (any) |
~50% |
70.8% |
61.2% |
~55% |
| Dysaesthesia |
Not reported |
22.9% |
6.0% |
Not reported |
| Discontinuation due to side effects |
~7% |
5.4% |
~7% |
~10% |
The STEP UP trial reported that 22.9% of people on Wegovy 7.2 mg experienced dysaesthesia, including tingling, numbness, or other abnormal skin sensations. This is higher than the 6% seen with the 2.4 mg dose.
However, 85.7% of these cases resolved while continuing treatment, and most were mild to moderate.2
Serious but rare side effects
Pancreatitis occurs in fewer than 0.2% of users but requires immediate medical attention if you experience severe abdominal pain.
Gallbladder problems, including gallstones, affect 1 to 2% of users, particularly with rapid weight loss.
Changes in diabetic retinopathy may occur in people with existing diabetes and require monitoring.
Cost analysis
Updated private prescription costs (2026)
| Medication |
Starting dose |
Maintenance dose |
Estimated annual cost |
| Mounjaro |
~£149/month (2.5 mg) |
~£300–375/month (15 mg) |
£3,600–4,500 |
| Wegovy 2.4 mg |
~£130/month (0.25 mg) |
~£200–295/month |
£2,400–3,540 |
| Wegovy 7.2 mg |
N/A (step-up from 2.4 mg) |
~£250–300/month (estimated) |
£3,000–3,600 |
| Saxenda |
~£100/month |
~£150–300/month |
£1,800–3,600 |
Prices vary by provider. Additional costs may include consultations (£50–150), blood tests (£50–100), and delivery charges.
Before September 2025, Mounjaro and Wegovy were priced similarly, making Mounjaro the more obvious choice given its higher weight-loss results.
With Mounjaro’s price now higher and Wegovy 7.2 mg available, Wegovy has become more attractive for many people.
It achieves 20.7% weight loss at an estimated £250 to £375 a month, while Mounjaro’s highest dose achieves 22.5% at £300 to £400 a month.
For many people, the additional weight loss with Mounjaro may not justify the higher cost over a 12- to 24-month treatment period, with the total difference ranging from £600 to £1,800.
This is a decision to make with your prescriber, based on your circumstances, budget, and your body’s response to each medication.
How to choose the right weight-loss injection
Practical decision factors
If you need maximum weight loss and the budget is not a constraint, Mounjaro at the 15 mg dose has the highest average results in clinical trials.
If cost is a concern and you want comparable results, Wegovy 7.2 mg achieves close to Mounjaro’s results at a lower price.
If you’re already on Wegovy 2.4 mg and have plateaued, stepping up to 7.2 mg may be worth discussing with your prescriber.
If you prefer daily dosing or need the lowest cost, Saxenda remains an option, though its results are substantially lower than the other two.
Medical considerations
Your prescriber will assess contraindications (reasons not to prescribe the medication), including thyroid cancer risk, a history of pancreatitis, and kidney function. Current medications need review.
If you take the oral contraceptive pill, be aware of the MHRA’s warning about Mounjaro potentially reducing its effectiveness.
If you’re planning a pregnancy, all GLP-1 medications must be stopped 2 to 3 months before conception.
Second Nature has worked with the NHS since 2017, providing Mounjaro and Wegovy alongside the habit-change support that helps weight loss last. Take our 3-minute eligibility quiz, and a clinician will review your answers.
Weight-loss medications in development
Several medications in late-stage trials may reach the UK within the next few years.
Retatrutide: the triple agonist
Eli Lilly’s retatrutide mimics three hormones: GLP-1, GIP, and glucagon. That is one more than Mounjaro, which mimics two.
In TRIUMPH-1, the first large Phase 3 trial (a large human trial with thousands of participants) in adults with obesity, retatrutide produced an average weight loss of 28.3% at the 12 mg dose after 80 weeks. Eli Lilly reported these topline results in 2026, ahead of full peer-reviewed publication.14
Almost half of the participants on the 12 mg dose lost 30% or more of their body weight, a level usually associated with weight-loss surgery.
An earlier Phase 3 trial in people with obesity and knee osteoarthritis, TRIUMPH-4, found an average weight loss of 28.7% at the same dose, alongside a reduction in joint pain.15
Further TRIUMPH trials in people with type 2 diabetes and cardiovascular disease are due to be released later in 2026.
If the results hold, retatrutide could be submitted to regulators in late 2026 or 2027, with UK availability most likely in 2027 to 2028.
A notable safety signal is dysaesthesia, which refers to tingling, numbness, or altered skin sensations, reported in about 1 in 5 participants at the 12 mg dose.
Discontinuation rates were higher than those for the current weekly injections, partly because some people lost more weight than they intended.
Oral semaglutide (the Wegovy pill)
Novo Nordisk launched an oral form of semaglutide for weight loss in the US in January 2026.
In the OASIS-4 trial, the once-daily 25 mg tablet produced an average weight loss of 13.6% over 64 weeks among all participants who started the trial.
Among people who stayed on it and reached the full dose, average weight loss was 16.6%.
The MHRA is reviewing the oral form, with a decision expected towards the end of 2026. If approved, it would suit people who would rather take a tablet than inject.
CagriSema
Novo Nordisk’s CagriSema combines semaglutide with cagrilintide, an amylin analogue, thereby mimicking two hormones rather than one.
In the REDEFINE 1 trial, people who stayed on treatment lost an average of 22.7% of their body weight.
Novo Nordisk submitted CagriSema to the FDA in December 2025, with a decision expected in late 2026 and UK availability likely in 2027 or later.
Orforglipron (Foundayo): a daily pill from Eli Lilly
The FDA approved orforglipron, branded Foundayo, for weight management in April 2026.
It’s the first GLP-1 medication available as a daily tablet that can be taken at any time of day, without the food-and-water timing rules required by oral semaglutide.
In its Phase 3 trials, orforglipron produced an average weight loss of around 12%.
The MHRA hasn’t yet approved it, so it is not available in the UK, though a UK submission is expected.
Frequently asked questions
Which weight-loss injection is most effective in the UK?
Mounjaro (tirzepatide) at the 15 mg dose has the highest average results in clinical trials, with 22.5% weight loss after 72 weeks.
The newer Wegovy 7.2 mg dose achieves 20.7%, a difference of around 1 to 2 percentage points.
The best choice for you depends on how your body responds, your budget, and your overall health.
Is Wegovy 7.2 mg as effective as Mounjaro?
Almost. Mounjaro 15 mg gave an average weight loss of 22.5% in SURMOUNT-1, and Wegovy 7.2 mg gave 20.7% in STEP UP, a difference of around 1 to 2 percentage points.
One in three people on Wegovy 7.2 mg lost 25% or more of their body weight, a level previously seen only with Mounjaro and with weight-loss surgery.2
Which weight-loss injection is right for me?
The best choice depends on how much weight you want to lose, your budget, whether you prefer a weekly or daily injection, and your overall health.
Mounjaro offers the largest average weight loss; Wegovy is often cheaper and is the only one with heart-health benefits on its label; and Saxenda suits people who prefer daily dosing.
Why has Mounjaro’s price increased?
Eli Lilly raised Mounjaro’s UK list price from September 2025 to bring it closer to pricing in other countries.
When Mounjaro launched in the UK in 2024, it was priced lower than in other European markets to support faster access.
The highest dose (15 mg) saw the largest increase, from £122 to £330 per four-week pen.
What is the cheapest weight-loss injection in the UK?
Since Mounjaro’s price increase in September 2025, Wegovy 2.4 mg has often been the cheaper option, at around £150 to £250 a month privately, compared with £300 to £375 a month for Mounjaro 15 mg.
Saxenda can be cheaper still, but its average weight loss is much lower.
Can I switch from Mounjaro to Wegovy to save money?
Yes, switching is possible with your prescriber’s guidance.
A 7-day washout period is usually recommended, and you would typically start at the relevant Wegovy dose based on your current Mounjaro dose.
Your prescriber can advise on the safest way to make this transition.
Can I get weight-loss injections on the NHS?
NHS access has expanded but remains limited.
Mounjaro became available through primary care in June 2025 as part of a phased rollout, initially for people with a BMI of 40 or above and at least four weight-related health conditions.
Wegovy 2.4 mg is available through specialist Tier 3 services with 6 to 24 months’ waiting times. Most people currently access these medications privately.
Can you buy weight-loss injections over the counter in the UK?
No. Mounjaro, Wegovy, and Saxenda are all prescription-only and require a medical assessment by a qualified prescriber.
Since January 2026, prescribers can no longer rely on an online questionnaire alone and must verify their weight, height, and BMI.
Is Saxenda still available in the UK?
The medication is still available, but the original Saxenda brand from Novo Nordisk has had long-term supply problems and is being phased out.
Since liraglutide’s patent expired in 2025, the same drug is now sold as generic liraglutide by other manufacturers, including Biocon’s Biolide and the brand Nevolat.
If you’re prescribed liraglutide today, it is likely to be one of these rather than branded Saxenda.
What happens when I stop taking weight-loss injections?
If you stop a weight-loss injection suddenly without having built the habits needed to maintain your new weight, some weight regain is likely.
A 2026 meta-analysis in the BMJ found that people regained a significant proportion of their lost weight within a year of stopping.12
Earlier data from the STEP 1 trial extension showed people regained roughly two-thirds of the weight they had lost within 12 months.13
This pattern of regain is consistent across trials that have examined stopping these medications abruptly.
We recommend coming off slowly, gradually reducing the dose while establishing habits to manage hunger naturally.
Our free weight regain prevention planner walks you through how to do this.
What’s the difference between Ozempic and Wegovy?
Both contain the drug semaglutide, but at different doses and for different purposes. Wegovy is approved for weight loss at doses up to 7.2 mg.
Ozempic is approved for type 2 diabetes at a maximum of 2 mg. Ozempic is not licensed for weight loss in the UK.
How quickly will I see results?
Most people notice reduced appetite within the first week or two.
Visible weight loss typically begins around weeks 4 to 8. Mounjaro and Wegovy users often see 5% weight loss by week 12, with maximum results at 12 to 18 months.
Does Mounjaro affect the contraceptive pill?
The MHRA has warned that Mounjaro may reduce the effectiveness of oral contraceptives.
If you take the combined pill, use an additional method such as condoms, or discuss switching to a non-oral contraceptive (patches, IUD, or injection) with your GP.
When will the Wegovy pill be available in the UK?
The oral form of semaglutide (25 mg tablet) launched in the US in January 2026.
The MHRA is reviewing it, with a decision expected towards the end of 2026. NHS availability would require a further review by NICE.
When will retatrutide be available in the UK?
Retatrutide is still in Phase 3 trials. Its first large obesity trial, TRIUMPH-1, reported topline results in 2026, with further trials due later in the year.
If all goes well, regulatory submission could follow in late 2026 or 2027, with UK availability most likely in 2027 to 2028.
Are weight-loss injections safe for long-term use?
Current safety data support long-term use with appropriate monitoring. The longest studies span 2 to 4 years with reassuring safety profiles.
Regular monitoring should include kidney function, gallbladder health, and thyroid checks.
For many people, the long-term cardiovascular and metabolic benefits of maintaining a healthy weight outweigh the risks of continued medication.
Take home message
Mounjaro remains the most effective weight-loss injection in the UK in 2026, with an average weight loss of 22.5% at the maximum dose. But the options available have changed considerably since 2025.
The MHRA approved Wegovy 7.2 mg in January 2026, which produced an average weight loss of 20.7%, close to Mounjaro’s trial figures.
At the same time, Mounjaro’s price increase from September 2025 means Wegovy is now often the more cost-effective option.
NHS access to Mounjaro has begun through primary care, but eligibility criteria are strict, and the rollout is phased over 12 years.
Most people will continue to access these medications privately for the foreseeable future.
All three available options, Mounjaro, Wegovy, and Saxenda, require a prescription following a proper medical assessment.
They work best when combined with sustainable changes to eating, exercise, and lifestyle habits. Medication on its own is unlikely to support lasting weight loss.
Research shows that people tend to regain weight if they stop suddenly without having built the habits needed to keep it off.
The support you receive alongside the medication, and the habits you build during treatment, matter as much as the medication itself.
At Second Nature, we provide Mounjaro and Wegovy alongside support from a registered dietitian and a structured habit-change programme built around the balanced plate.
Our published research found members lost an average of 19.1% of their body weight at 12 months, with 77.7% losing at least 10%.8
Second Nature's Mounjaro and Wegovy programmes
Second Nature provides Mounjaro or Wegovy as part of our Mounjaro and Wegovy weight-loss programmes.
Why choose Second Nature over other medication providers, assuming you're eligible?
Because peace of mind matters.
We've had the privilege of working with the NHS for over eight years, helping people across the UK take meaningful steps toward a healthier, happier life.
Our programmes are designed to meet people where they are, whether that means support with weight loss through compassionate one-to-one health coaching, or access to the latest weight-loss medications (like Mounjaro and Wegovy) delivered alongside expert care from a multidisciplinary team of doctors, psychologists, dietitians, and personal trainers.
At the heart of everything we do is a simple belief: real, lasting change comes from building better habits, not relying on quick fixes. We're here to support that change every step of the way.
With over a decade of experience, thousands of lives changed, and a long-standing record of delivering programmes used by the NHS, we believe we're the UK's most trusted weight-loss programme.
We hope to offer you something invaluable: peace of mind, and the support you need to take that first step.






