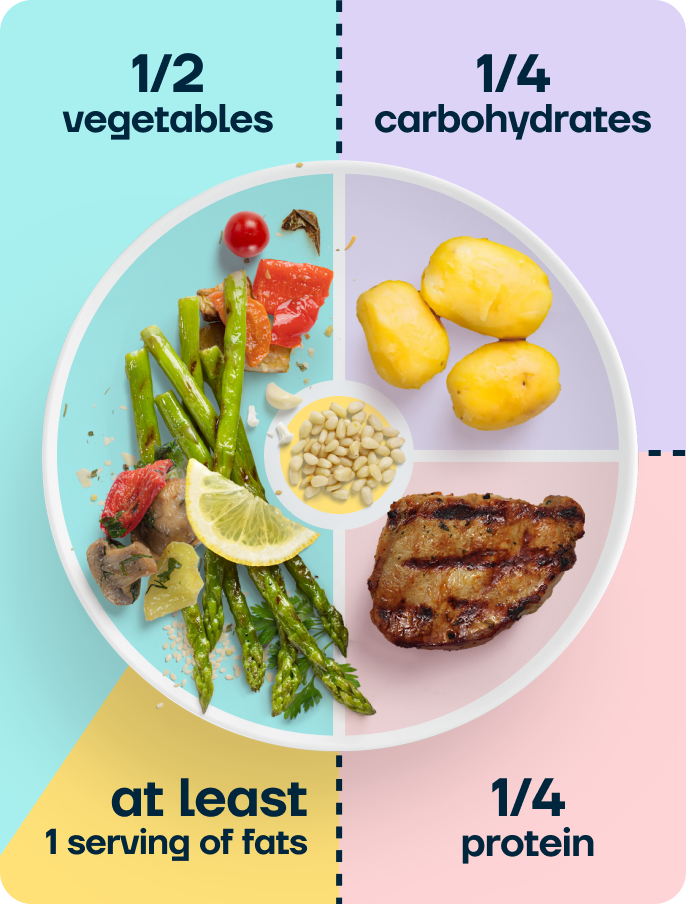When it’s safe to start
Wegovy isn’t recommended during active cancer treatment. The SELECT trial and other major obesity studies excluded people with a recent cancer diagnosis.
The gastrointestinal side effects of semaglutide (nausea, vomiting, reduced appetite) can overlap with chemotherapy and radiotherapy side effects, making symptoms harder to manage.2
Most oncologists prefer people to have finished active treatment (surgery, chemotherapy, radiotherapy) and to be in documented complete remission before starting Wegovy.
In a 2026 cohort of breast cancer survivors at MD Anderson Cancer Center, the median time from breast cancer diagnosis to starting a GLP-1 receptor agonist was around four years.3
Being on long-term endocrine therapy (tamoxifen, anastrozole, letrozole, or exemestane) doesn’t prevent you from taking Wegovy.
The same cohort followed survivors who started GLP-1 medications while on endocrine therapy and reported improved overall survival, with no increase in disease-free survival events.3
Questions to bring to your oncologist
- Is my remission status documented as ‘no evidence of disease’?
- Do you have any concerns about starting a GLP-1 medication now?
- Am I due for any scans or tests soon that should happen first?
- Are any of my current medications likely to interact?
- How will we monitor me once I’ve started?
Safety evidence
The clinical evidence so far doesn’t suggest Wegovy increases the risk of cancer recurrence.
The SELECT trial tested semaglutide 2.4 mg in over 17,000 people with cardiovascular disease and obesity over an average of 3.3 years.2 A 2025 safety analysis compared cancer rates between the semaglutide and placebo groups.
Breast cancer occurred in 0.7% of women on semaglutide compared with 0.8% on placebo. Pancreatic and thyroid cancers were equally rare in both groups.2
A 2025 meta-analysis of 50 randomised trials of GLP-1 receptor agonists found no increased overall cancer risk compared with placebo.
A small increase in thyroid cancer risk appeared across the class, though the absolute risk remains very low; no increase in risk was found for breast cancer. Uterine cancer was reduced in the obesity trials.4
Laboratory studies on breast cancer cell lines (cancer cells grown in a dish) have shown mixed effects of GLP-1 medications, but cell-line research doesn’t translate directly into human outcomes.
Wegovy compatibility with breast cancer treatments
| Treatment |
Pharmacokinetic interaction |
Timing recommendation |
| Tamoxifen |
None expected; different metabolic pathways |
Can start while on long-term therapy |
| Aromatase inhibitors (anastrozole, letrozole, exemestane) |
None expected; different metabolic pathways |
Can start while on long-term therapy; weight loss may be slower |
| CDK4/6 inhibitors (palbociclib, abemaciclib, ribociclib) |
None expected; semaglutide doesn’t use CYP3A4 |
Confirm with oncologist; clinical interaction data limited |
| Trastuzumab (Herceptin), Kadcyla |
None expected; different clearance routes |
Inform the oncology team |
| Chemotherapy |
No PK interaction, but overlapping GI side effects |
Wait until active treatment is complete |
| Radiotherapy |
No PK interaction, but overlapping fatigue and GI symptoms |
Wait until active treatment is complete |
Interactions with cancer treatments
Semaglutide is broken down by enzymes that digest proteins, not by the liver enzymes (cytochrome P450) that process most medications.5 This means it doesn’t compete with drugs that are processed by the liver.
Tamoxifen and aromatase inhibitors
Tamoxifen is activated by liver enzymes into its active form, endoxifen. Because semaglutide doesn’t use these enzymes, no interaction is expected, and none has been reported.
Aromatase inhibitors are processed through different pathways from semaglutide. No drug-drug interaction is expected.
Weight loss with Wegovy is often slower while on endocrine therapy.
In the MD Anderson cohort, semaglutide and tirzepatide users had a median weight change of -2.6% at 12 months, smaller than the figures typically seen in general obesity trials.3
CDK4/6 inhibitors
Palbociclib, abemaciclib, and ribociclib are processed by the CYP3A4 liver enzyme.
Because semaglutide doesn’t affect this enzyme, no interaction is expected. Published clinical interaction data is limited, so confirm with your oncology team before starting.
Trastuzumab and Kadcyla
These biologic therapies are given by infusion and cleared through different routes from semaglutide.
No pharmacokinetic interaction has been reported. Tell your oncology team about any new medication, regardless.
Chemotherapy and radiotherapy
Wegovy isn’t recommended during active chemotherapy or radiotherapy.
The combined gastrointestinal effects make side effects difficult to separate and manage, and weight loss during treatment can interfere with chemotherapy dosing.
Weight loss and breast cancer recurrence
Carrying excess weight after a breast cancer diagnosis is associated with worse outcomes.
A meta-analysis of 82 studies involving over 213,000 survivors found that women with obesity had a 41% higher risk of dying from any cause compared with women at a healthy weight.6
Whether deliberately losing weight reduces the risk of recurrence is a more complicated question.
The Women’s Intervention Nutrition Study (WINS), a randomised controlled trial in over 2,400 early-stage breast cancer survivors, tested a low-fat dietary intervention that produced modest weight loss.
Women in the intervention group had a 24% lower relapse rate after five years.7
The Breast Cancer Weight Loss (BWEL) trial tested a structured weight-loss programme in 3,180 women with stage II or III breast cancer.
At one year, intervention participants had lost 4.7% of their body weight compared with a 1% gain in the control group.8
Recurrence data from BWEL haven’t been published yet.
The World Cancer Research Fund’s 2025 review concluded that the evidence on deliberate weight loss improving breast cancer outcomes isn’t yet definitive.9
What is well established is that weight loss improves cardiovascular health, reduces the side effects of endocrine therapy, and improves physical function, all of which matter during cancer treatment.
Why endocrine therapy often brings weight gain
Many women gain weight after a breast cancer diagnosis, and endocrine therapy contributes.
Aromatase inhibitors and tamoxifen are associated with shifts in body composition, with reductions in lean muscle mass and increases in fat mass, even when overall weight stays roughly the same.
Treatment-related fatigue, the joint pain that affects up to half of women on aromatase inhibitors, and reductions in physical activity all add to this.10
Post-treatment weight gain isn’t a personal failing. It reflects real biological and treatment-related changes that affect almost every woman in this position.
Bone health on aromatase inhibitors
Bone density is a particular concern after breast cancer. Aromatase inhibitors accelerate bone loss, and women on long-term AI therapy have a higher risk of osteoporosis and fragility fracture compared with the general postmenopausal population.11
Rapid weight loss of any kind is also associated with reduced bone density.
A 2024 randomised trial in adults with elevated fracture risk found that semaglutide reduced hip bone mineral density by 2.6% and lumbar spine density by 2.1% at 52 weeks compared with placebo.12
For women on aromatase inhibitors, the bone loss from AI therapy and the bone loss from rapid weight loss can compound.
If you’ve had a recent DEXA scan, share the results with your prescriber before starting Wegovy. If you haven’t had one, ask your oncology team whether one is due.
Three things help protect bone density during weight loss
- Adequate calcium and vitamin D. Most adults need around 700 mg of calcium daily from food and a 10 microgram vitamin D supplement, particularly through winter months in the UK.
- Resistance training twice a week, which directly stimulates bone formation
- Regular DEXA monitoring as recommended by your oncology team
If you’re already taking a bisphosphonate or denosumab for bone protection, continue taking them as prescribed.
Aromatase inhibitor joint pain
Joint pain is the most common reason women stop aromatase inhibitor therapy early, and stopping AI treatment ahead of schedule is associated with higher recurrence risk.10
The HOPE trial randomised 121 breast cancer survivors with AI-related arthralgia to a year-long exercise programme of aerobic activity plus supervised strength training, or usual care.
Women in the exercise group reported significant reductions in joint pain compared with usual care, with the largest benefit appearing after 12 months of regular exercise.10
Weight loss may add further benefit. Excess weight increases the load on weight-bearing joints, and joint pain in osteoarthritis improves with both weight loss and exercise.
There’s no specific trial yet of GLP-1 medications for AI-related joint pain, but the combination of structured exercise and gradual weight loss aligns with what’s already shown to help.
If AI joint pain is interfering with daily life, this is worth raising with your oncologist alongside the question of Wegovy.
Cardiovascular health
Many breast cancer survivors are at elevated cardiovascular risk. Anthracycline chemotherapy (doxorubicin, epirubicin) and left-sided radiotherapy can affect cardiac function years after treatment ends.
The SELECT trial showed that semaglutide 2.4 mg reduced major adverse cardiovascular events by 20% in people with overweight or obesity and established cardiovascular disease, and benefits appeared early, before substantial weight loss had occurred.2
These results don’t mean Wegovy is appropriate purely as a cardioprotective medication, and breast cancer survivors weren’t specifically studied in SELECT.
But for someone whose oncology team is also weighing post-treatment cardiovascular risk, the wider evidence base on semaglutide is reassuring.
Reconstruction timing
If you’re planning breast reconstruction, your weight needs to be stable before surgery.
Significant weight changes during or after reconstruction (whether DIEP flap, implant exchange, or fat grafting) can affect symmetry, fit, and aesthetic results.
Most plastic surgeons recommend reaching a stable weight before reconstruction surgery and avoiding major weight changes in the months afterwards.
If you’re considering Wegovy and reconstruction within the same year, discuss timing with both your oncology and plastic surgery teams before starting.
Lymphoedema considerations
Higher BMI at the time of breast cancer diagnosis is a well-established risk factor for breast cancer-related lymphoedema.
Women with a BMI of 30 or above have around three times the risk of those at a healthy weight.13
The relationship between weight loss after diagnosis and lymphoedema progression is less clear.
Some studies of dietary weight-loss interventions in survivors show benefit, others don’t.
Significant weight fluctuations (whether gain or loss) appear to increase risk more than steady, gradual change.13
If you have lymphoedema, gradual rather than rapid weight loss is preferable.
Continue compression garments and lymphatic drainage as prescribed, and let your lymphoedema specialist know if you’re starting Wegovy.
Foods to focus on
Eating well after breast cancer focuses on building an eating pattern that supports recovery, reduces inflammation, and protects muscle mass during weight loss.
A diet based on whole foods
A balanced plate of whole foods supports recovery, helps protect muscle mass during weight loss, and gives you the nutrients your body needs while healing.
In practice, this means filling roughly half your plate with vegetables, a quarter with protein (chicken, fish, eggs, beans, lentils, tofu, or Greek yoghurt), and a quarter with complex carbohydrates (wholegrain bread, brown rice, oats, potatoes, or quinoa). Add a portion of healthy fat, such as olive oil, avocado, nuts, or seeds.
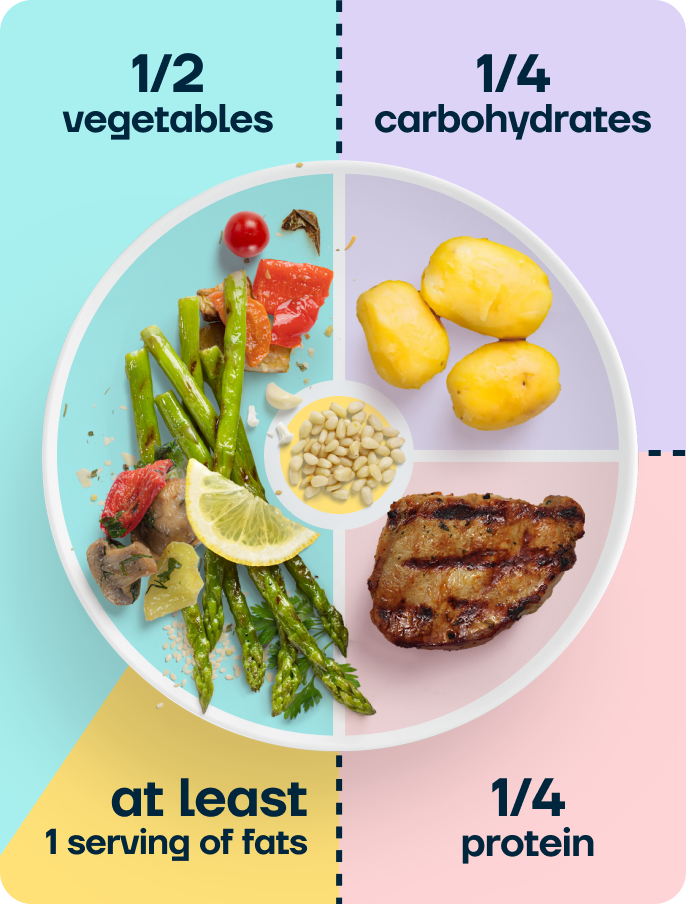
Build most of your meals around these foods. Ultra-processed foods (ready meals, biscuits, crisps, sugary drinks) are fine in smaller amounts, but they tend to be lower in protein, fibre, and the nutrients that support recovery, so they shouldn’t form the basis of your meals.
Protein at each meal
Getting enough protein matters more than usual when you’re losing weight, because it helps preserve muscle mass.
Muscle loss during treatment and weight loss can worsen fatigue and reduce physical function.
Good sources include chicken, fish, eggs, Greek yoghurt, and beans and lentils. We recommend a palm-sized portion at each meal.
Oily fish like salmon, mackerel, and sardines are particularly worth including for their omega-3 content.
Soy foods are safe
A common worry after hormone-receptor-positive breast cancer is whether to avoid soy.
Research across large cohorts shows that moderate intake of whole soy foods, such as tofu, edamame, tempeh, and soya milk, is safe and may be associated with a lower risk of recurrence, including in women taking aromatase inhibitors.14
Concentrated isoflavone supplements are a separate question with less evidence, so whole foods are the safer starting point.
Alcohol
Alcohol raises breast cancer risk and is associated with higher recurrence rates in survivors, particularly postmenopausal women.15
Breast Cancer Now recommends limiting alcohol to less than 5 units a week after a breast cancer diagnosis.
Alcohol also worsens the nausea and reflux that some people experience on Wegovy, which is another practical reason to keep intake low.
Staying active
Exercise has a strong evidence base supporting its use in cancer survival.
A 2023 analysis of post-diagnosis physical activity found a 16% reduction in recurrence and a 23% reduction in breast cancer-specific mortality in the most active survivors.16
Getting started
If you’re coming back from treatment, fatigue, deconditioning, and changes in body shape can all make exercise feel harder than it used to.
A 10-minute walk after dinner is a practical starting point. Building consistency over time is more useful than how hard any single session is.
Resistance training (including after lymph node surgery)
Older guidance discouraged lifting weights after axillary surgery, but current evidence shows resistance training doesn’t worsen lymphoedema and improves strength and shoulder function.
Starting light and progressing gradually is safer than avoiding strength work altogether.
Bodyweight exercises, resistance bands, or light dumbbells are all good starting points.
If you’ve had axillary node surgery, a lymphoedema specialist physiotherapist can guide the first few weeks.
Aerobic exercise
Walking, swimming, and cycling are all low-impact and easy to build into a routine.
Swimming can be particularly comfortable if you have joint pain from aromatase inhibitors, because water takes the weight off your joints.
Looking after your mental health
A breast cancer diagnosis and its treatment affect mental health in ways that can persist long after physical recovery.
A UK primary-care cohort study found that breast cancer survivors had a 33% higher risk of anxiety and a 35% higher risk of depression compared with women without a cancer history.17
The risk was highest in the first three years after diagnosis but remained elevated.
Body shape changes during weight loss can feel complicated after surgery or reconstruction, fear of recurrence may intensify when starting a new medication, and the fatigue that often lingers after treatment can make the early weeks of Wegovy feel harder.
Body image after treatment
Weight change after breast cancer treatment can affect how you feel about your body in ways that are more complex than weight loss alone.
Scars, reconstruction, changes in sensation, and lymphoedema can all affect how you feel about losing weight.
Cancer-specific psychological support is available through Macmillan Cancer Support (macmillan.org.uk) and Breast Cancer Now (breastcancernow.org). NHS Talking Therapies accepts self-referral in most areas of England.
Fear of recurrence
Starting a new medication can trigger fear of recurrence, particularly when online information about GLP-1s and cancer is confusing.
If that’s affecting you, speaking to your oncology clinical nurse specialist can be more grounding than searching online. Macmillan also runs a free support line on 0808 808 00 00.
When to speak to your GP or oncologist
Contact your GP, prescriber, or oncology team if you experience:
- Any new lumps, changes in the affected or unaffected breast, or new symptoms that prompt concern about recurrence
- Persistent nausea or vomiting that stops you from keeping fluids down
- Rapid or significant weight loss beyond what’s expected
- Signs of dehydration (dark urine, dizziness, reduced urination)
- New fatigue that feels different from your post-treatment fatigue
- Worsening joint pain or new bone pain
- Increasing anxiety, low mood, or fear of recurrence that’s affecting daily life
- Any difficulty attending cancer follow-up appointments because of side effects
Don’t delay routine cancer surveillance because of Wegovy. Mammograms, blood tests, and oncology follow-ups should continue as scheduled.
Frequently asked questions
How long after treatment should I wait before starting Wegovy?
Most oncologists prefer you to have finished active treatment (surgery, chemotherapy, radiotherapy) and have documented complete remission.
Real-world data from cohorts of breast cancer survivors shows a median of around four years between diagnosis and starting a GLP-1 medication, though the right timing for you depends on your treatment plan.3
Can I take Wegovy while on tamoxifen or anastrozole?
There’s no pharmacokinetic interaction between semaglutide and these medications.
Weight loss may be slower while you’re on endocrine therapy, but it may still provide benefit. Discuss with your oncologist before starting.3
Will Wegovy increase my risk of recurrence?
Current evidence doesn’t suggest it does. The SELECT safety analysis found breast cancer rates were marginally lower on semaglutide than placebo, and a 2025 meta-analysis of 50 randomised GLP-1 trials found no increased breast cancer risk.
A cohort study in survivors found improved overall survival and no increase in disease-free survival events.2,3,4
Will Wegovy reduce my risk of recurrence?
Evidence that weight loss reduces recurrence comes mainly from dietary intervention trials like WINS, not from GLP-1 medications specifically.7
The BWEL trial will provide stronger answers when long-term results are published.
The World Cancer Research Fund’s 2025 review rated the evidence on deliberate weight loss and breast cancer outcomes as uncertain rather than definitive.9
Does Wegovy affect my bones?
A 2024 randomised trial showed semaglutide reduced hip bone mineral density by 2.6% and lumbar spine density by 2.1% at 52 weeks compared with placebo, in adults already at increased fracture risk.12
If you’re on aromatase inhibitors (which also accelerate bone loss), discuss DEXA monitoring and bone-protective measures with your oncology team before starting.
Can Wegovy help with aromatase inhibitor joint pain?
There’s no specific trial of GLP-1 medications for AI-related joint pain.
The HOPE trial showed that exercise reduces AI joint pain in breast cancer survivors,10 and weight loss is known to reduce joint loading.
The combination of structured exercise plus gradual weight loss is reasonable to discuss with your oncologist if joint pain is affecting AI adherence.
I’m planning breast reconstruction. Should I lose weight first?
Most plastic surgeons recommend reaching a stable weight before reconstruction. Significant weight changes during or after surgery can affect aesthetic outcomes.
If you’re considering Wegovy and reconstruction in the same year, discuss timing with both teams before starting.
Is soy safe to eat if I’m on Wegovy after ER-positive breast cancer?
Yes, a moderate intake of whole soy foods is safe and may be associated with lower recurrence rates, even in women on aromatase inhibitors.14
Tofu, edamame, tempeh, and soya milk are fine. Concentrated isoflavone supplements are less well studied.
Does Wegovy affect mammogram or blood test results?
There’s no published evidence that semaglutide interferes with mammographic imaging or tumour markers like CA 15-3. Continue your surveillance as scheduled.
I’m on a CDK4/6 inhibitor like palbociclib or abemaciclib. Can I take Wegovy?
No pharmacokinetic interaction is expected, because semaglutide doesn’t affect the liver enzymes that process CDK4/6 inhibitors.
Research on these combinations is limited, so confirm with your oncologist before starting.
What if I’m still losing weight from treatment rather than gaining?
Wegovy isn’t usually appropriate if you’re underweight or have lost significant weight from treatment.
The priority will more likely be to focus on building healthy habits to support recovery, overall health, and regain muscle mass.
Can I get Wegovy on the NHS after breast cancer?
NICE recommends semaglutide for weight management in eligible adults through specialist weight management services (TA875).18
A history of cancer isn’t an explicit exclusion, but eligibility depends on your BMI, current health, and local service criteria.
NHS specialist weight management services are also limited and have long waiting lists in many areas, so private prescribing is often more accessible. Your GP can advise on local options.
Take home message
Most breast cancer survivors in complete remission can take Wegovy.
The Wegovy UK prescribing information has no cancer-related contraindications, and a 2025 SELECT trial analysis found no increase in breast cancer rates on semaglutide compared with placebo.1,2
Most oncologists prefer that you have completed active treatment and be on stable follow-up.
Being on tamoxifen or an aromatase inhibitor doesn’t prevent you from taking Wegovy, though weight loss may be slower than in general obesity trials.3
Whether weight loss reduces the risk of recurrence remains an open question.
What’s better established is that weight loss improves cardiovascular health, may reduce aromatase inhibitor joint pain, and supports overall physical function during survivorship.
Alongside the medication, monitor bone density if you’re on aromatase inhibitors, lose weight gradually if you have lymphoedema, and keep your weight stable before any planned reconstruction.
Second Nature’s programme combines medication support with personalised nutrition guidance from registered dietitians and nutritionists, built around a balanced plate of vegetables, protein, complex carbohydrates, and healthy fats.
A peer-reviewed study published in JMIR Formative Research found that active subscribers on Second Nature’s semaglutide-supported programme lost an average of 19.1% of their body weight at 12 months, with 77.7% achieving at least 10% weight loss.19
Second Nature's Mounjaro and Wegovy programmes
Second Nature provides Mounjaro or Wegovy as part of our Mounjaro and Wegovy weight-loss programmes.
Why choose Second Nature over other medication providers, assuming you're eligible?
Because peace of mind matters.
We've had the privilege of working with the NHS for over eight years, helping people across the UK take meaningful steps toward a healthier, happier life.
Our programmes are designed to meet people where they are, whether that means support with weight loss through compassionate one-to-one health coaching, or access to the latest weight-loss medications (like Mounjaro and Wegovy) delivered alongside expert care from a multidisciplinary team of doctors, psychologists, dietitians, and personal trainers.
At the heart of everything we do is a simple belief: real, lasting change comes from building better habits, not relying on quick fixes. We're here to support that change every step of the way.
With over a decade of experience, thousands of lives changed, and a long-standing record of delivering programmes used by the NHS, we believe we're the UK's most trusted weight-loss programme.
We hope to offer you something invaluable: peace of mind, and the support you need to take that first step.
References
- Electronic Medicines Compendium. (2026). Wegovy 2.4 mg FlexTouch solution for injection in pre-filled pen: Summary of Product Characteristics.
- Kushner, R.F. et al. (2025). Safety profile of semaglutide versus placebo in the SELECT study: a randomized controlled trial. Obesity.
- Sukumar, J.S. et al. (2026). Weight loss patterns and clinical outcomes of GLP1 receptor agonists in breast cancer survivors. Cancer Research Communications, 6(3), 447-455.
- Silverii, G.A. et al. (2025). GLP-1 receptor agonists and the risk for cancer: a meta-analysis of randomised controlled trials. Diabetes, Obesity and Metabolism, 27(8), 4454-4468.
- Hausner, H. et al. (2017). Effect of semaglutide on the pharmacokinetics of metformin, warfarin, atorvastatin and digoxin in healthy subjects. Clinical Pharmacokinetics, 56(11), 1391-1401.
- Chan, D.S.M. et al. (2014). Body mass index and survival in women with breast cancer: a systematic review and meta-analysis of 82 follow-up studies. Annals of Oncology, 25(10), 1901-1914.
- Chlebowski, R.T. et al. (2006). Dietary fat reduction and breast cancer outcome: interim efficacy results from the Women’s Intervention Nutrition Study. Journal of the National Cancer Institute, 98(24), 1767-1776.
- Ligibel, J.A. et al. (2025). Impact of a weight loss intervention on 1-year weight change in women with stage II/III breast cancer: secondary analysis of the BWEL trial. JAMA Oncology, 11(10), 1194-1203.
- World Cancer Research Fund. (2025). Diet, nutrition, physical activity and body weight for people living with and beyond breast cancer. CUP Global Report.
- Irwin, M.L. et al. (2015). Randomized exercise trial of aromatase inhibitor-induced arthralgia in breast cancer survivors (HOPE study). Journal of Clinical Oncology, 33(10), 1104-1111.
- Hadji, P. et al. (2025). Management of aromatase inhibitor-associated bone loss in women with hormone-sensitive breast cancer: an updated joint position statement of the IOF, CABS, ECTS, IEG, ESCEO, IMS, and SIOG. Journal of Bone Oncology.
- Hansen, M.S. et al. (2024). Once-weekly semaglutide versus placebo in adults with increased fracture risk: a randomised, double-blinded, two-centre, phase 2 trial. eClinicalMedicine.
- Lin, Y. et al. (2020). Lymphoedema after breast cancer treatment is associated with higher body mass index: a systematic review and meta-analysis. International Journal of Cancer.
- Shu, X.O. et al. (2009). Soy food intake and breast cancer survival. JAMA, 302(22), 2437-2443.
- Kwan, M.L. et al. (2010). Alcohol consumption and breast cancer recurrence and survival among women with early-stage breast cancer: the Life After Cancer Epidemiology Study. Journal of Clinical Oncology, 28(29), 4410-4416.
- Morze, J. et al. (2023). Postdiagnosis recreational physical activity and breast cancer prognosis. International Journal of Cancer.
- Carreira, H. et al. (2021). Associations between breast cancer survivorship and adverse mental health outcomes: a matched population-based cohort study in the UK. PLOS Medicine, 18(1), e1003504.
- NICE. (2023). Semaglutide for managing overweight and obesity. Technology appraisal TA875.
- Richards, R. et al. (2025). A remotely delivered GLP-1RA-supported specialist weight management program in adults living with obesity: retrospective service evaluation. JMIR Formative Research, 9, e72577.